News
2026-04-23
Master Thesis Antonio Alvarez
|
Antonio Alvarez submitted his Master thesis on reproducible Bayesian optimization and uncertainty quantification in pharmacokinetic models /paper/theses/Master.Thesis.Antonio.Alvarez.pdf Antonio Alvarez developed a reproducible Bayesian framework for parameter estimation in pharmacokinetic (PK) and physiologically-based pharmacokinetic (PBPK) models. Using three models of increasing complexity, he systematically evaluated how key hyperparameters influence Bayesian MCMC performance on synthetic datasets. The study demonstrates that prior specification is the dominant factor for accurate parameter recovery, while other settings mainly affect computational cost. The fully open and FAIR-compliant workflow provides a robust foundation for uncertainty quantification and reproducible PBPK modeling. |
2026-03-20
Preprint: A Digital Twin of Empagliflozin Pharmacokinetics and Pharmacodynamics
|
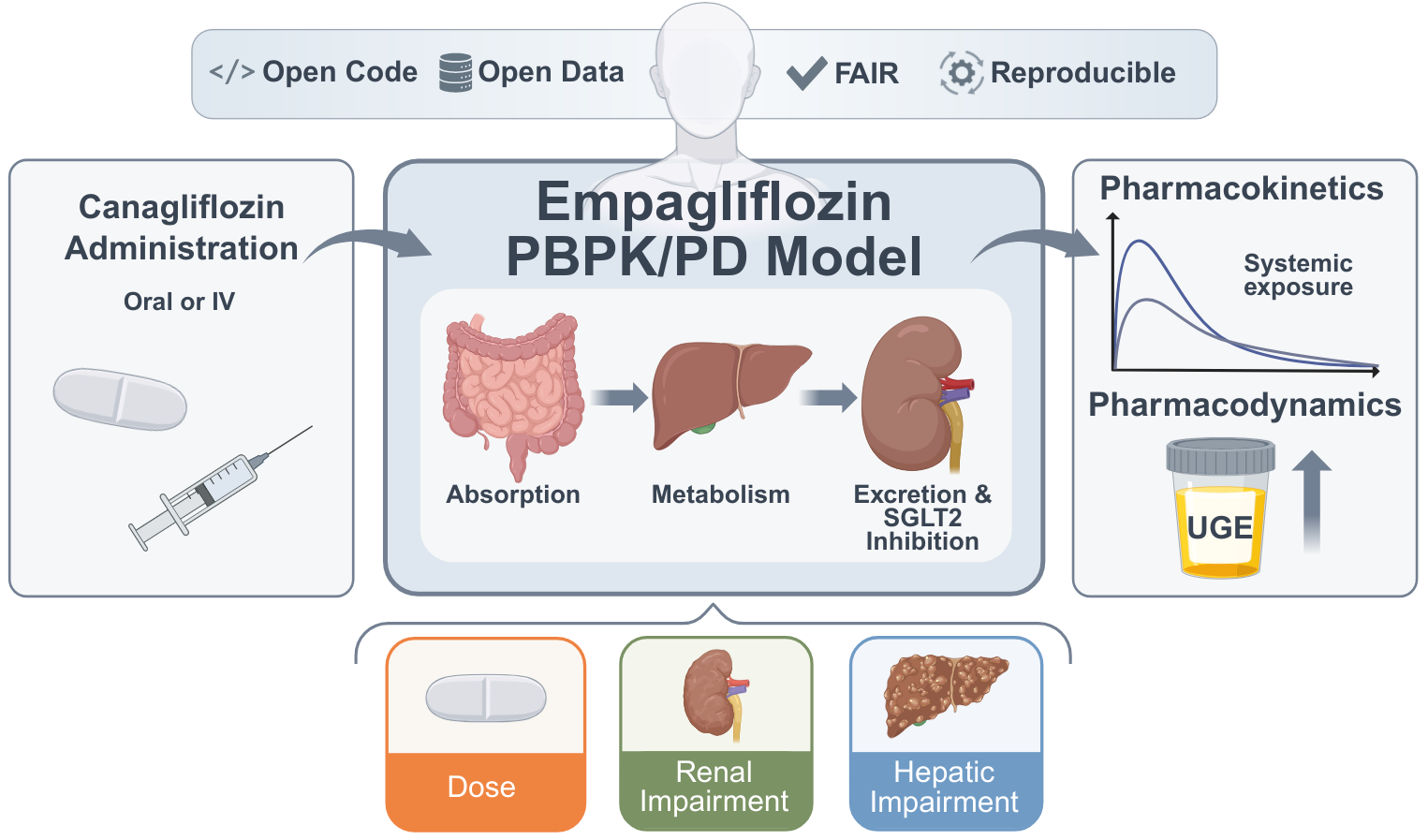
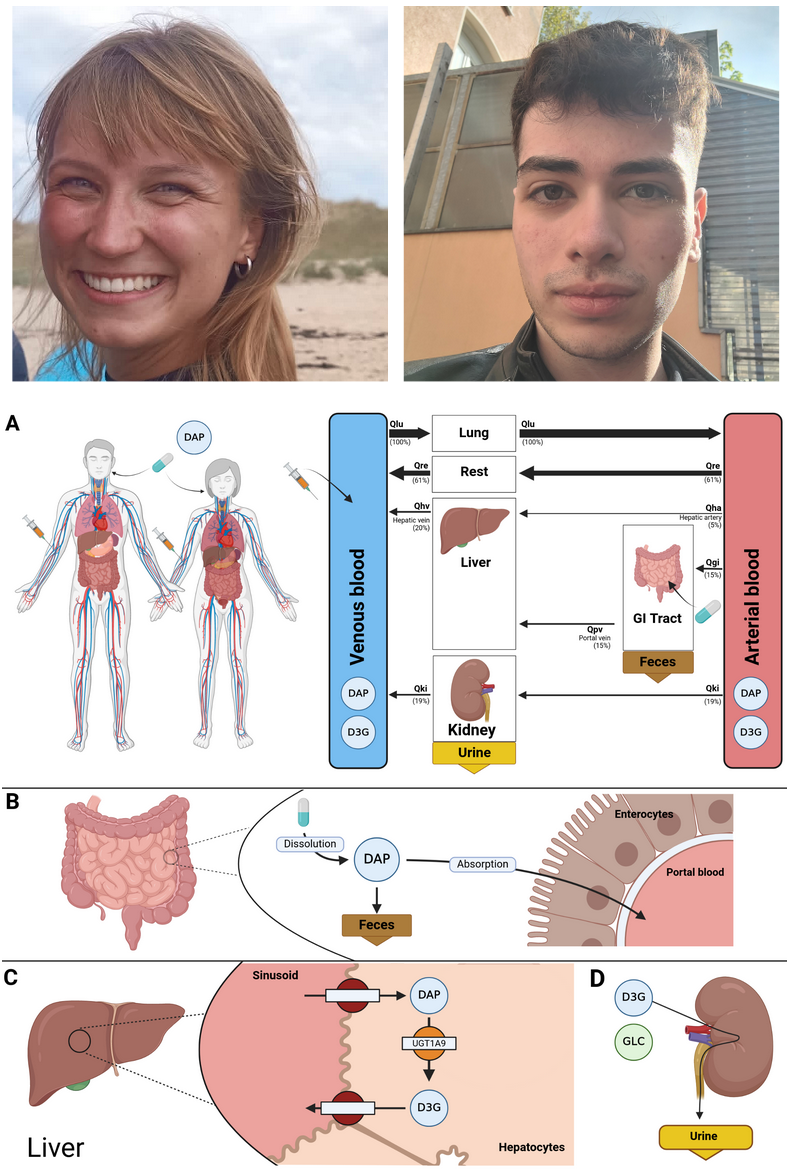
We present a whole-body PBPK/PD digital twin of empagliflozin to investigate pharmacokinetic and pharmacodynamic variability in type 2 diabetes mellitus, with a focus on renal and hepatic impairment, metabolite exposure, and dose-dependent effects. https://www.preprints.org/manuscript/202603.1559/v1 In a new preprint, Jennesse Alejandro, Michelle Elias, Mariia Babaeva, and Matthias König introduce a physiologically based PBPK/PD digital twin of the SGLT2 inhibitor empagliflozin, developed using curated data from 27 clinical studies. The whole-body model mechanistically represents absorption, distribution, metabolism, and excretion, and explicitly links drug exposure to renal glucose handling via the renal threshold for glucose and urinary glucose excretion. Simulations reproduce dose-dependent pharmacokinetics and pharmacodynamics across single- and multiple-dose regimens in healthy individuals and patients with type 2 diabetes. The model captures variability under renal and hepatic impairment as well as fed and fasted conditions, providing quantitative insight into clinically relevant scenarios. All models, datasets, and simulation workflows are provided as open, FAIR-compliant SBML resources, enabling reproducible analyses and reuse for model-informed assessment of empagliflozin PK/PD variability. |
2026-03-12
Keynote: Digital Twins and Dashboards for Personalized Drug Treatment
|
|
At the Patient Monitoring Roundtable (PMRT) at Charité – Universitätsmedizin Berlin, I presented how digital twins and interactive dashboards can support personalized drug treatment and clinical decision-making. Digital twins are emerging as a powerful concept in data-driven and personalized medicine. At the Patient Monitoring Roundtable (PMRT) hosted at Charité – Universitätsmedizin Berlin, I gave a keynote on how mechanistic computational models and interactive dashboards can be used to simulate individualized drug responses and support clinical decision-making. The talk introduced digital twins in pharmacokinetics based on physiologically based pharmacokinetic (PBPK) models, which represent human physiology in silico and simulate how drugs are absorbed, distributed, metabolized, and eliminated. Using examples such as digital twins of glimepiride and liver function models, I showed how patient-specific factors—including genetics, liver function, renal impairment, and body weight—can influence drug exposure and treatment outcomes. Interactive dashboards allow clinicians and researchers to explore these models intuitively, enabling scenario testing, risk prediction, and shared decision-making with patients. The session was followed by interactive workshops where participants discussed digital twins and dashboards. Many thanks to the organizers and fellow speakers for a fantastic event and inspiring discussions on the future of data-driven healthcare. |
2026-01-27
Preprint: A Digital Twin of Canagliflozin Pharmacokinetics and Pharmacodynamics in Type 2 Diabetes Mellitus
|
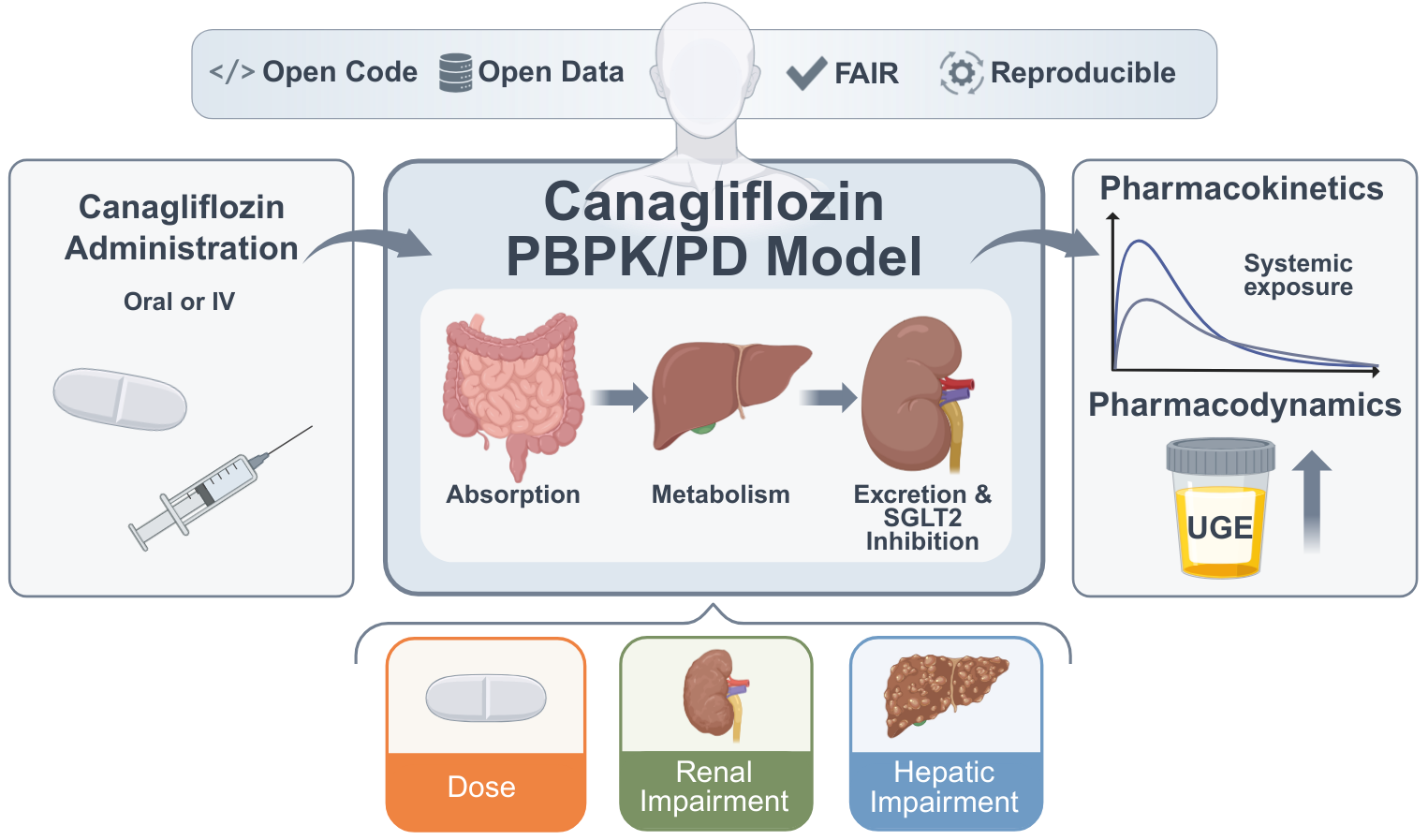
We present a whole-body PBPK/PD digital twin of canagliflozin to investigate pharmacokinetic and pharmacodynamic variability in type 2 diabetes mellitus, with a focus on renal and hepatic impairment, metabolite exposure, and dose-dependent effects. https://www.preprints.org/manuscript/202601.2095/v1 In a new preprint, Vera Tereshchuk, Michelle Elias, and Matthias König introduce a physiologically based PBPK/PD digital twin of the SGLT2 inhibitor canagliflozin, developed using curated data from 22 clinical studies. The whole-body model mechanistically represents absorption, distribution, metabolism, and excretion, and explicitly links drug exposure to renal glucose handling and urinary glucose excretion. Simulations reproduce dose-dependent pharmacokinetics and pharmacodynamics across single- and multiple-dose regimens in healthy individuals and patients with type 2 diabetes. Renal impairment results in modest changes in parent drug exposure but markedly reduced urinary glucose excretion and increased metabolite exposure, consistent with impaired glucose filtration and clearance. Under hepatic impairment, simulations predict increased canagliflozin exposure with altered metabolite profiles, while pharmacodynamic effects remain minimal. All models, datasets, and simulation workflows are provided as open, FAIR-compliant SBML resources, enabling reproducible analyses and reuse for model-informed assessment of canagliflozin PK/PD variability. |
2026-01-30
Interview: Modelle so komplex wie der Körper selbst – Digitale Zwillinge in der Medizin
|
In an interview with Matthias König, the Gesellschaft für Informatik explores how digital twins are becoming as complex as the human body itself and how they enable personalized prediction, simulation, and decision support in medicine. https://inf.gi.de/12/modelle-so-komplex-wie-der-koerper-selbst In this interview published by the Gesellschaft für Informatik, Matthias König discusses the concept of digital twins in medicine and how increasingly detailed computational models can reflect the complexity of the human body across scales. The interview highlights the role of systems biology, physiologically based pharmacokinetic and pharmacodynamic modeling, and data-driven approaches in building predictive digital twins for organs and whole-body physiology. Key application areas include personalized therapy optimization, drug development, and clinical decision support. The conversation also addresses current challenges such as model validation, data integration, interoperability, and reproducibility, emphasizing the importance of open, FAIR, and community-driven modeling standards for the future of precision and systems medicine. |
2025-11-28
Successful Open Science Workshop 2025

|
The Open Science Workshop 2025 brought together participants from across the Berlin University Alliance for a full-day, hands-on introduction to reproducible research, FAIR data, and open-source workflows. https://matthiaskoenig.github.io/open-science-workshop The Open Science Workshop 2025 was a great success, welcoming a diverse group of participants from all BUA universities. The workshop covered key aspects of open and reproducible research, including FAIR data principles, version control with Git, transparent workflows, open-source tools, computational reproducibility, and best practices for collaborative science. All workshop materials—including slides, hands-on exercises, and code examples—are openly available on the workshop website (https://matthiaskoenig.github.io/open-science-workshop), with full source files and datasets provided in the public GitHub repository (https://github.com/matthiaskoenig/open-science-workshop). A detailed reflection and recap of the workshop, including participant demographics and learning outcomes, is available here: https://matthiaskoenig.github.io/open-science-workshop/docs/workshop2025.html The workshop fostered engaging discussions on the future of open science and highlighted the importance of transparency, accessibility, and reproducibility in modern research. Participants reported a significant improvement in their understanding of all presented technologies. Certificates of participation were provided to all attendees. |
2025-11-20
Completion of the AI Teaching Certificate (KI-Lehrzertifikat)

|
Successful completion of the AI Teaching Certificate (200 AE) at Humboldt-Universität zu Berlin, covering AI foundations, ethics, and higher education didactics within the KI-Campus 2.0 and AI Skills Initiative. Matthias König has successfully completed the AI Teaching Certificate (KI-Lehrzertifikat), a comprehensive 200-unit higher education qualification program offered through KI-Campus 2.0 and the AI Skills Initiative at Humboldt-Universität zu Berlin. The certificate consists of three micro-credentials—AI Foundations (96%), AI Advanced & Ethics (93%), and AI Didactics in Higher Education (91%)—and covers core AI topics including machine learning, neural networks, Python and Jupyter workflows, text and image processing, and responsible AI use. Training followed DGHD-aligned standards in AI ethics, data and algorithmic governance, and regulatory aspects. The program also emphasized AI-supported teaching, such as integrating generative AI, speech assistants, and prompt design into university courses, as well as developing AI-based OER teaching units. The certification was completed with a teaching observation and reflective assessments demonstrating competency in designing and implementing AI-enhanced learning scenarios. |
2025-11-13
Humboldt Internship Program 2026 - Computational Modeling of Drug Detoxification Using Digital Twins – A Systems Medicine Approach

|
We offer Internships in 2026, academic three-month research stays at Humboldt-Universität zu Berlin. The application for the Summer Term Internships 2026 is open from 05 November to 18 December 2025. https://hic.hu-berlin.de/en/internship-program/projects/201
Humboldt Internship Program is an international short-term program for subject-specific, experiential learning. It allows participants to work with teams in research projects and university-related start-ups for three months. |
2025-11-13
A Digital Twin of Dapagliflozin Pharmacokinetics and Pharmacodynamics in Type 2 Diabetes Mellitus

|
We present a physiologically based PBPK/PD digital twin of dapagliflozin to analyze pharmacokinetic and pharmacodynamic variability in type 2 diabetes mellitus, with a focus on dosing, hepatorenal impairment, and food effects. https://github.com/matthiaskoenig/dapagliflozin-model In a new preprint, Nike Nemitz, Michelle Elias, and Matthias König introduce a digital twin of the SGLT2 inhibitor dapagliflozin, developed as a whole-body PBPK/PD model based on curated data from 28 clinical studies. The model mechanistically describes the drug’s absorption, distribution, metabolism, excretion, and pharmacodynamic action via urinary glucose excretion. It captures key sources of inter-individual variability—including renal and hepatic impairment, food effects, and dose-dependent kinetics—and successfully reproduces observed clinical PK/PD patterns. Simulations reveal that renal impairment substantially reduces urinary glucose excretion despite preserved plasma exposure, while hepatic impairment has only minor influence. Food intake lowers peak concentrations but still leads to measurable changes in pharmacodynamic response. This digital twin provides mechanistic insight into dapagliflozin variability and supports individualized therapy optimization in type 2 diabetes. |
2025-11-01
Faces of the Berlin University Alliance
|
|
I am very honored to be featured in the new film series launched for the 6th anniversary of the Berlin University Alliance (BUA). Watch my short portrait: 🎥. Learn more about the Berlin University Alliance: 🎥. https://www.youtube.com/playlist?list=PLNPRvG2dBEvvWlnLg0C3WPtTYAOOH110k
I am very honored to be featured in the new film series launched for the 6th anniversary of the Berlin University Alliance (BUA) — the research alliance of FU Berlin, HU Berlin, TU Berlin, and Charité – Universitätsmedizin Berlin. The series highlights researchers who work across institutions, disciplines, and fields to shape Berlin as a place of knowledge and innovation. It shows how collaboration drives new ideas — from molecular research to cultural studies, from clinical practice to digital infrastructures. In my portrait, I talk about our work on digital twins of the liver — computational models that integrate physiology, pharmacokinetics, and clinical data to support personalized medicine, teaching, and open science. |
2025-10-14
Digital Twin of Losartan for Precision Blood Pressure Therapy

|
We introduce a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of losartan and its active metabolite E3174 as a digital twin to study variability in blood pressure regulation and support individualized antihypertensive therapy. https://github.com/matthiaskoenig/losartan-model In a new preprint, Ennie Tensil and Matthias König present a digital twin of the angiotensin II receptor blocker losartan, providing a mechanistic framework for precision treatment of hypertension and heart failure. The whole-body PBPK/PD model, based on data from 24 clinical trials, describes losartan’s absorption, hepatic metabolism, renal excretion, and pharmacodynamic effects via the renin–angiotensin–aldosterone system (RAAS). Implemented in SBML, the model captures interindividual variability arising from hepatic and renal impairment as well as CYP2C9 and ABCB1 polymorphisms. Simulations reproduce key pharmacokinetic and pharmacodynamic patterns, highlighting the impact of genetic and physiological factors on therapeutic response. This digital twin enables virtual patient studies and provides a quantitative foundation for individualized dosing and precision cardiovascular therapy. |
2025-10-20
COMBINE & RDA: Reproducibility by Design: Data and Workflows for Pharmacokinetic Digital Twins
|
|
Presenting at COMBINE2025 and the RDA assembly our approach to reproducible digitial twins. https://github.com/matthiaskoenig/glimepiride-model
Dr. Matthias König (Humboldt-Universität zu Berlin) presents how reproducibility and open standards enable trustworthy Digital Twins in pharmacology and medicine. Using the example of the glimepiride digital twin for type 2 diabetes, he demonstrates:
- How patient-specific data, genetics, and physiology shape individualized drug responses - How open standards (SBML, SED-ML, PEtab, OMEX) ensure interoperability - How Executable Simulation Models (EXSIMO) combine model, data, and code for full transparency - Independent reproducibility checks by the Physiome Journal and BioModels Team
Explore more: |
2025-10-08
Digital Twin of Glimepiride Enables Personalized Diabetes Therapy

|
We present a digital pharmacokinetic twin of glimepiride that simulates how patient-specific factors affect drug behavior, supporting individualized and stratified treatment of type 2 diabetes. https://github.com/matthiaskoenig/glimepiride-model In a new publication, Michelle Elias and Matthias König introduce a whole-body physiologically based pharmacokinetic (PBPK) model of glimepiride as a digital twin for precision diabetes care. Developed using curated data from 19 clinical studies, the model captures glimepiride’s ADME processes and quantitatively integrates key sources of interindividual variability—including hepatic and renal function, CYP2C9 genotype, and bodyweight. The digital twin accurately reflects observed pharmacokinetics and demonstrates how genetic and physiological factors can significantly alter drug exposure. By enabling virtual patient simulations, this model offers a mechanistic platform for personalized dosing and patient stratification, advancing digital decision-support tools for more effective and individualized treatment of type 2 diabetes. |
2025-09-16
Automated Segmentation of Hepatic Vessels and Lobules in Whole-Slide Images Using U-Net Models

|
Our latest preprint introduces a deep-learning pipeline for automated segmentation of hepatic vessels, bile ducts, and lobules in whole-slide liver histology images. https://doi.org/10.1101/2025.09.08.674181 Automated analysis of hepatic vascular structures and lobules in histological whole-slide images is essential for computational liver histology. In this preprint, Bafna, König, Saalfeld, Moulisova, Liska, Dahmen, and Albadry present a robust deep-learning pipeline based on U-Net models with adaptive patch extraction and weight-boosted nnU-Net architectures. The framework accurately segments lobules (Dice 0.960), central veins (0.801), hepatic arteries (0.909), portal veins (0.609), and bile ducts (0.710) in stained porcine liver sections, while supporting morphometric analyses of structural features. The study demonstrates the potential of adaptable segmentation models to improve precision, reproducibility, and scalability in computational histopathology. |
2025-09-15
Assessing the Impact of AI and Digital Twins on Clinical Decision-Making in Hepatology and Hepatobiliary Surgery

|
Our latest preprint presents results from a survey study exploring how clinicians in hepatology and hepatobiliary surgery perceive artificial intelligence (AI) and digital twins in clinical decision-making, highlighting both enthusiasm for innovation and concerns about trust, liability, and regulation. https://doi.org/10.20944/preprints202509.1164.v1 Artificial Intelligence (AI) and Digital Twins (DT) are poised to transform clinical decision-making in hepatology and hepatobiliary surgery, yet their adoption depends on clinician trust and regulatory clarity. We conducted an online survey among 18 specialists, including hepatologists, hepatobiliary surgeons, and related clinicians, to assess familiarity, perceived value, adoption of clinical decision support systems (CDSS), concerns about automation bias and liability, and attitudes toward patient-centred integration. Results showed high perceived value of AI (80%) and DT (79%), with 93% reporting current or intended CDSS use. However, 71% of respondents expressed concern over automation bias, and 92% were uncertain about legal protection. Importantly, 77% supported integrating patient preferences into AI- and DT-assisted decision-making. Our findings reveal strong interest in AI and DT adoption but also significant caution, underscoring the need for clear guidelines, clinical validation, and patient-centred frameworks to ensure safe and effective integration into clinical practice. |
2025-08-15
Leadership Certificate Successfully Completed by Dr. Matthias König

|
Dr. Matthias König has successfully completed the comprehensive Leadership Certificate of the Berlin Leadership Academy, strengthening his leadership skills for guiding research teams and fostering effective collaboration in academia. Dr. Matthias König has successfully completed the Leadership Certificate for Junior Professors and Junior Research Group Leaders at the Berlin Leadership Academy (BLA) of the Berlin University Alliance. This intensive, two-stage program supports participants who are taking on leadership responsibilities for the first time, focusing on the specific challenges of leadership in academia and higher education. Through a combination of workshops, interactive exercises, and case-based discussions, the program covered strategies for effective collaboration, structured organization of research teams, constructive communication across hierarchical levels, and maintaining personal health and resilience in leadership roles. By completing this program, Dr. König has further strengthened his leadership and social competencies, preparing him to guide his research group and collaborative projects with greater impact and confidence. |
2025-07-15
Bachelor Thesis Ennie Tensil: Physiologically-Based Pharmacokinetic/Pharmacodynamic Modeling of Losartan

|
This thesis presents a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of losartan and its metabolite E3174, providing mechanistic insights into how physiological and genetic factors influence therapy and supporting individualized dosing strategies. https://github.com/matthiaskoenig/losartan-model Losartan is an angiotensin II receptor antagonist commonly used in the treatment of hyperten- sion and heart failure. Its pharmacokinetics and pharmacodynamics are influenced by a range of physiological and genetic factors, which may affect therapeutic efficacy. This thesis presents a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of losartan and its active metabolite E3174 to explore these influences systematically. The modular SBML-based model includes submodels for absorption, hepatic metabolism, renal excretion, and pharmacodynamic effects via the renin-angiotensin-aldosterone system (RAAS). Simulations were conducted to investigate the impact of dose, hepatic and renal impairment, as well as CYP2C9 and ABCB1 polymorphisms. The model successfully reproduced observed pharmacological trends, including a dose-dependent increase in pharmacodynamic response, likely reflecting enhanced receptor blockade at higher systemic exposures. Hepatic dysfunction blunted the pharmacodynamic response to losartan, whereas renal impairment reduced metabolite clearance and lead to modest enhancement in pharmacodynamic effect. Genetic variability in CYP2C9 significantly altered E3174 formation and downstream RAAS inhibition, whereas ABCB1 activity had only minor effects on systemic exposure. The model provides mechanistic insight into inter-individual variability in losartan therapy and supports its potential use in individualized dosing strategies. |
2025-07-14
A Systems Pharmacology Approach to Rivaroxaban: Physiologically Based Modeling of Pharmacokinetics and Coagulation Dynamics

|
Our latest preprint introduces a comprehensive physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of rivaroxaban, enabling the simulation of drug absorption, distribution, metabolism, excretion, and coagulation dynamics under diverse clinical conditions. https://github.com/matthiaskoenig/rivaroxaban-model Rivaroxaban, a widely used direct oral anticoagulant (DOAC), shows significant inter-individual variability in pharmacokinetics and pharmacodynamics, especially in patients with altered physiology or comorbidities. In this study, we established an open database of PK/PD data from 14 clinical studies and developed a mechanistic whole-body PBPK/PD model to describe rivaroxaban’s behavior under varying doses, food intake conditions, and levels of hepatic and renal impairment. The model accurately reproduces observed plasma concentration-time profiles and coagulation dynamics, including Factor Xa inhibition, prothrombin time (PT), and activated partial thromboplastin time (aPTT). By capturing key sources of variability, the model provides a robust framework to explore personalized dosing strategies, improve clinical decision-making, and enable integration into digital twin platforms and clinical decision support systems. |
2025-07-07
Humboldt Internship Program Program Day 2025: Project Report & Panel Discussion

|
Isabella Tan, Olivia Yau and Minjun Kim presented there PBPK/PD modeling projects on GLP-1 agonists for Program Day here at Humboldt Universität-zu Berlin. https://github.com/matthiaskoenig/losartan-model During Program Day at Humboldt-Universität zu Berlin, Isabella Tan, Olivia Yau, and Minjun Kim presented their physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) modeling projects on GLP-1 receptor agonists. Minjun Kim is developing a model of dulaglutide to investigate its absorption, distribution, metabolism, and effects on glucose regulation. Isabella Tan is building a model of liraglutide to study its pharmacological behavior and impact on glucose homeostasis, focusing on individualized treatment strategies. Olivia Yau is working on a model of lixisenatide to explore its kinetics and therapeutic effects, aiming to advance personalized approaches to diabetes therapy. Additionally, Matthias König contributed as a panelist to the discussion 'Real Work Experiences for a Real Career: What Learnings and Skills Really Matter?' |
2025-07-02
New PBPK/PD Modeling Project: Rapamycin as a Case Study in Personalized Immunosuppressive Therapy

|
Monika Jesionek has started a new ERASMUS project to develop a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of rapamycin, aiming to better understand interindividual variability and support the development of an immune digital twin. As part of her ERASMUS research stay, Monika Jesionek is developing a whole-body PBPK/PD model of the mTOR inhibitor rapamycin (sirolimus). Rapamycin plays a crucial role in immunosuppressive therapy, but its clinical use is challenged by narrow therapeutic margins, complex pharmacokinetics, and high interindividual variability. The project integrates literature data and mechanistic modeling to simulate rapamycin’s absorption, distribution, metabolism, and immune-modulating effects across diverse physiological and pathological conditions. This digital twin approach aims to optimize dosing strategies and contribute to personalized medicine efforts, particularly within the broader context of building an immune digital twin (IDT). |
2025-06-17
Digital Twin of Glimepiride Enables Personalized Diabetes Therapy

|
We present a digital pharmacokinetic twin of glimepiride that simulates how patient-specific factors affect drug behavior, supporting individualized and stratified treatment of type 2 diabetes. https://github.com/matthiaskoenig/glimepiride-model In a new preprint, Michelle Elias and Matthias König introduce a whole-body physiologically based pharmacokinetic (PBPK) model of glimepiride as a digital twin for precision diabetes care. Developed using curated data from 19 clinical studies, the model captures glimepiride’s ADME processes and quantitatively integrates key sources of interindividual variability—including hepatic and renal function, CYP2C9 genotype, and bodyweight. The digital twin accurately reflects observed pharmacokinetics and demonstrates how genetic and physiological factors can significantly alter drug exposure. By enabling virtual patient simulations, this model offers a mechanistic platform for personalized dosing and patient stratification, advancing digital decision-support tools for more effective and individualized treatment of type 2 diabetes. |
2025-06-15
Bachelor Thesis Nike Nemitz: Physiologically-Based Pharmacokinetic/Pharmacodynamic Modeling of Dapagliflozin

|
In this bachelor thesis, a PBPK/PD model of dapagliflozin was developed to investigate how physiological and external factors such as hepatorenal impairment and food intake influence drug behavior and therapeutic efficacy in type 2 diabetes treatment. https://github.com/matthiaskoenig/dapagliflozin-model Dapagliflozin, an SGLT2 inhibitor for type 2 diabetes, reduces plasma glucose levels by enhancing urinary glucose excretion (UGE). However, its pharmacokinetics and pharmacodynamics are subject to considerable interindividual variability influenced by dosing, organ function, and food intake. This study presents a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model, constructed using SBML and data from 27 clinical trials, with modular representations of intestinal absorption, hepatic metabolism, and renal excretion. The model reliably reproduces observed dose-dependent PK/PD profiles and simulates physiological variability and food effects. Simulations reveal that renal impairment markedly reduces UGE, while hepatic impairment has a minor impact. Food intake significantly lowers peak plasma concentrations (Cmax) without affecting AUC, highlighting a clinically relevant effect on pharmacodynamics often overlooked. This work provides mechanistic insight into key determinants of dapagliflozin variability and supports its use in predictive modeling for individualized therapy in diabetes care. |
2025-06-12
Panel Reflections on Inclusion in Research Design

|
Dr. Matthias König joined an interdisciplinary panel at the Berlin University Alliance Fellows Club to discuss how inclusive design influences research across fields. The event highlighted diverse approaches—from AI in healthcare to quantum computing—to advance pluralistic and equitable science. Methods of Inclusion: Designing Research for a Plural World” — part of the Berlin University Alliance Fellows Club meets... the Einstein Center Population Diversity on June 11. Together with an inspiring group of researchers from diverse fields—ranging from sociology to quantum computing—we explored how inclusion, representation, and plurality shape research design across disciplines. I had the opportunity to share perspectives from our work on AI-driven healthcare and digital twins, and how inclusive design principles are essential for building fair and transparent decision-support tools in medicine. A big thank you to the organizers from Berlin University Alliance, Einstein Center Population Diversity, and Stiftung Charité for creating a space for cross-institutional exchange and thoughtful dialogue. #Diversity #DigitalTwins #InclusiveResearch #BerlinUniversityAlliance #PopulationDiversity #SystemsMedicine #OpenScience. Thanks to Camilla Leathem, PhD, Sarah Bellows-Blakely, Michaela Kreyenfeld, Günter M. Ziegler, Lara Bister, Cole Kovarik and Fariha Azad |
2025-06-12
Course Completed: Pharmacokinetic/Pharmacodynamic Modelling

|
From June 3–6, we successfully conducted our redesigned course on Pharmacokinetic/Pharmacodynamic (PK/PD) Modelling. Developed as part of the Digital Health Profession Educator (DHPE) program, the course emphasized interactivity and hands-on learning in the context of clinical pharmacology. https://matthiaskoenig.github.io/dhpe-pkpd/ The Pharmacokinetic/Pharmacodynamic (PK/PD) Modelling course was completely restructured for enhanced interactivity as part of a teaching innovation project within the Digital Health Profession Educator (DHPE) program. The course provided a comprehensive introduction to how drugs are absorbed, distributed, metabolized, and excreted in the body—and how these processes affect drug action. Students explored both classical compartment models and physiologically based pharmacokinetic (PBPK) models, learning how to simulate drug behavior and optimize dosing strategies. The redesigned format integrated active learning components, practical modeling exercises, and real-world case studies to deepen understanding. Topics also included pharmacodynamics, drug-drug interactions, and the application of models in drug development and personalized therapy. The course successfully combined conceptual knowledge with applied skills, preparing participants to use PK/PD models as tools for translational and clinical research. |
2025-05-10
SPP Startup Funding Awarded for Web-Based FEM Visualization Tool

|
Matthias König has been awarded €15,000 in startup funding from the DFG Priority Programme SPP2311 to develop SPP-FEMVis, a web-based visualization platform for Finite Element Method (FEM) simulations supporting open science and interdisciplinary collaboration. We are pleased to announce that Matthias König has received startup funding from the DFG Priority Programme SPP2311 – Robust Coupling of Continuum Biomechanical In Silico Models to Biomedical Data. His project, SPP-FEMVis: Advancing Open Science with Web-Based FEM Visualizations, aims to create an interactive, browser-based visualization tool for FEM simulation results using PyVista and Trame. With €15,000 in funding, the project will support the development of a flexible visualization framework integrated into websites and Jupyter Notebooks, enabling seamless sharing and exploration of simulation data. The tool will increase accessibility and reproducibility of computational results across biomedical applications, including liver modeling, heart mechanics, and vascular simulations. By lowering technical barriers and fostering collaborative use of FEM models, SPP-FEMVis will strengthen the open science mission of SPP2311 and serve as a dissemination bridge between researchers, clinicians, and the public. |
2025-05-10
Algonomy at Festival della Robotica 2025: AI and Digital Twins for Liver Surgery

|
At the Festival della Robotica 2025 in Pisa, Mariia Myshkina presented our research from the Algonomy project, highlighting how AI and Digital Twins can support decision-making in hepatobiliary surgery. The Algonomy project, which investigates the ethical and practical integration of AI in clinical decision-making, was featured at the Festival della Robotica 2025 in Pisa. Representing the team, Mariia Myshkina presented our latest research on Digital Twins in hepatobiliary surgery—demonstrating how computational models of liver anatomy and function, powered by AI, can enhance surgical planning and individualized treatment strategies. Our contribution emphasized the central mission of Algonomy: combining algorithmic intelligence with clinical expertise while ensuring transparency, reproducibility, and shared decision-making in medicine. |
2025-05-10
New Projects Launched: Modeling GLP-1 Agonists and Optimizing Anesthesia Control

|
We’re excited to announce new research projects in our group focusing on personalized treatment strategies for diabetes and improved anesthesia management through computational modeling and control systems. Kim, Isabella, and Olivia have launched new projects developing physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) models of the GLP-1 receptor agonists dulaglutide, liraglutide, and lixisenatide, respectively. Their work aims to enhance our understanding of these drugs’ behavior and effects on glucose regulation, contributing to more individualized approaches to diabetes treatment. In parallel, Juan Pablo is working on a closed-loop control system for optimizing anesthesia delivery, with the goal of improving precision and patient safety in clinical settings. |
2025-04-30
Interview: Matthias König on Reproducibility, Open Science, and the Future of Biological Research

|
In an interview for the Berlin University Alliance, Matthias König discusses the reproducibility crisis in biology and the transformative potential of Open Science. As a BUA Open Science Ambassador, he highlights the importance of FAIR data, open standards, and cultural change in academia. On April 30, 2025, Matthias König, BUA Open Science Ambassador, was interviewed about the role of Open Science in improving reproducibility and collaboration in biological research. Drawing from his work on computational modeling and digital twins, König outlines how open data, FAIR principles, and transparent code are essential for reproducible science. He points to systemic barriers—such as incentive structures focused solely on publications—and calls for alternative evaluation criteria that recognize Open Science engagement. He also emphasizes the importance of community-driven initiatives, institutional support, and education to build a more open and collaborative research culture. The interview underscores the critical role of Open Science in enabling responsible AI development and long-term progress in life sciences. |
2025-04-15
Bachelor Thesis Michelle Elias: PBPK Modelling of Glimepiride: Investigating Variability in Type 2 Diabetes Treatment

|
Michelle Elias has successfully completed her Bachelor thesis on a physiologically based pharmacokinetic (PBPK) model of glimepiride, focusing on interindividual variability, hepatorenal impairment, and pharmacogenomics. https://github.com/matthiaskoenig/glimepiride-model Glimepiride, a widely used sulfonylurea for type 2 diabetes, shows significant interindividual variability in pharmacokinetics due to factors such as organ function and genetic polymorphisms. In her thesis, Michelle Elias developed a modular, SBML-based PBPK model using data from 19 clinical studies to simulate glimepiride absorption, hepatic CYP2C9-mediated metabolism, and renal excretion. The model accurately reproduced dose-dependent pharmacokinetics and revealed the distinct impacts of renal and hepatic impairment, as well as the influence of CYP2C9 genotypes and bodyweight. While limitations exist regarding data in severely impaired populations, the model offers mechanistic insights into variability and supports individualized dosing strategies for improved diabetes care. |
2025-04-01
SBML editor

|
Matthias König elected as an SBML Editor for the 2025–2027 term. Honored to be elected as an SBML Editor for the 2025–2027 term alongside Brett Olivier. Grateful for the trust of the community and excited to contribute to the future of SBML. A big thank you to outgoing editors Sarah Keating and Rahuman Sheriff for their dedicated work! #SBML #SystemsBiology #ComputationalModeling #OpenScience |
2025-04-02
Featured Fellow - Berlin University Alliance (BUA) - Matthias König

|
As a featured BUA Fellow at Humboldt-Universität, Matthias König advances digital medicine and systems biology at the intersection of data, modeling, and clinical research. Meet Matthias König, BUA Fellow and systems biologist at Humboldt-Universität zu Berlin. He develops computational models—so-called digital twins—that simulate human metabolism and organ function to advance personalized medicine. With a background in physics and systems medicine, Matthias leads research that integrates mathematical modeling, bioinformatics, and clinical data to better understand liver and kidney function. His work contributes to national and European efforts to build data-driven, predictive tools for healthcare. When asked about Berlin’s research landscape, he says: “Berlin offers a unique mix of scientific freedom and collaboration. It’s an exciting place for interdisciplinary research that really makes a difference.” |
2025-02-21
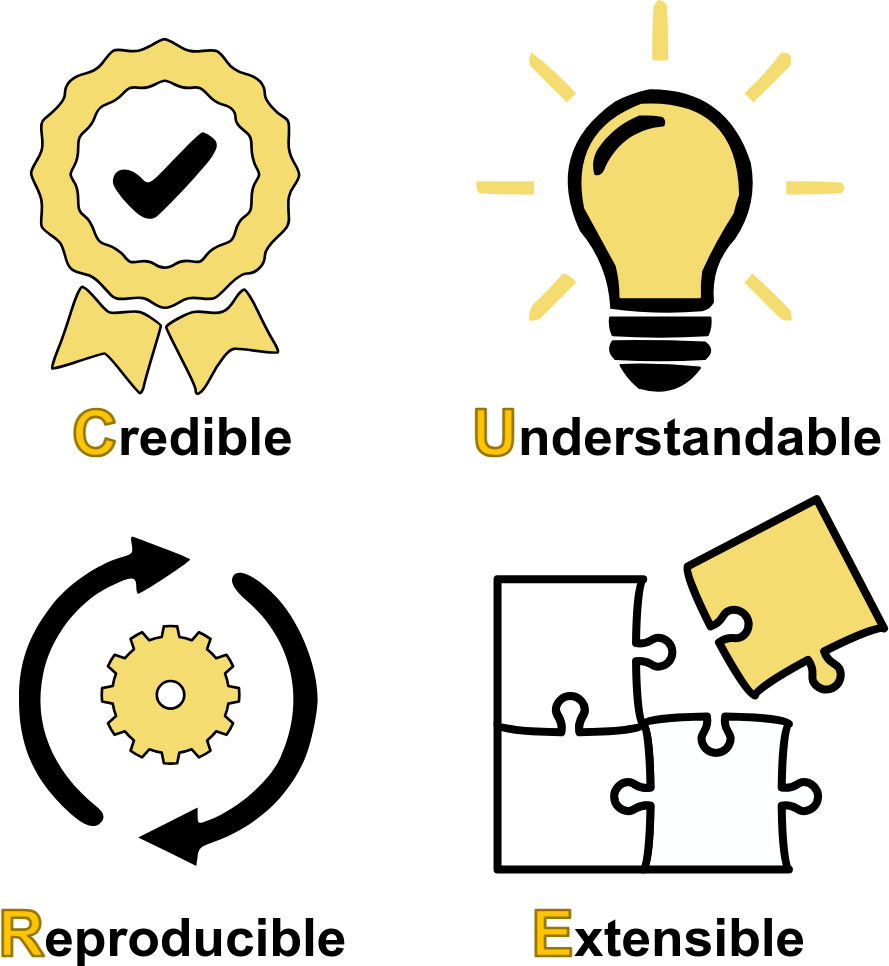
Preprint: From FAIR to CURE: Guidelines for Computational Models of Biological Systems
|
Our latest preprint, From FAIR to CURE: Guidelines for Computational Models of Biological Systems, introduces the CURE principles—Credible, Understandable, Reproducible, and Extensible—as a framework to improve the development and use of computational models in biology and medicine. https://doi.org/10.48550/arXiv.2502.15597/ Guidelines for managing scientific data have been established under the FAIR principles requiring that data be Findable, Accessible, Interoperable, and Reusable. In many scientific disciplines, especially computational biology, both data and models are key to progress. For this reason, and recognizing that such models are a very special type of 'data', we argue that computational models, especially mechanistic models prevalent in medicine, physiology and systems biology, deserve a complementary set of guidelines. We propose the CURE principles, emphasizing that models should be Credible, Understandable, Reproducible, and Extensible. We delve into each principle, discussing verification, validation, and uncertainty quantification for model credibility; the clarity of model descriptions and annotations for understandability; adherence to standards and open science practices for reproducibility; and the use of open standards and modular code for extensibility and reuse. We outline recommended and baseline requirements for each aspect of CURE, aiming to enhance the impact and trustworthiness of computational models, particularly in biomedical applications where credibility is paramount. Our perspective underscores the need for a more disciplined approach to modeling, aligning with emerging trends such as Digital Twins and emphasizing the importance of data and modeling standards for interoperability and reuse. Finally, we emphasize that given the non-trivial effort required to implement the guidelines, the community moves to automate as many of the guidelines as possible. |
2025-02-12
König Lab at EP PerMed Conference on Personalised Medicine Research

|
We are excited to announce that Mariia Myshkina, Elisabetta Casabianca, and Matthias König participated in the EP PerMed Conference on Personalised Medicine Research, taking place on February 11–12, 2025, in Berlin, Germany. https://www.eppermed.eu/news-events/events/ep-permed-conference-on-personalised-medicine-research/ We are excited to announce that Mariia Myshkina, Elisabetta Casabianca, and Matthias König participated in the EP PerMed Conference on Personalised Medicine Research, taking place on February 11–12, 2025, in Berlin, Germany. This conference explores key advancements in personalised medicine, focusing on methodologies, translational research, personalised prevention, and inclusiveness in healthcare. Discussions will span diagnostics, therapy, big data, clinical trials, ethics, and more. The event provides a unique opportunity for multidisciplinary collaboration to advance the field and shape the future of personalised medicine. We look forward to engaging discussions, networking, and contributing to this important initiative! For more details, visit the EP PerMed website: https://www.eppermed.eu/news-events/events/ep-permed-conference-on-personalised-medicine-research/ |
2025-01-20
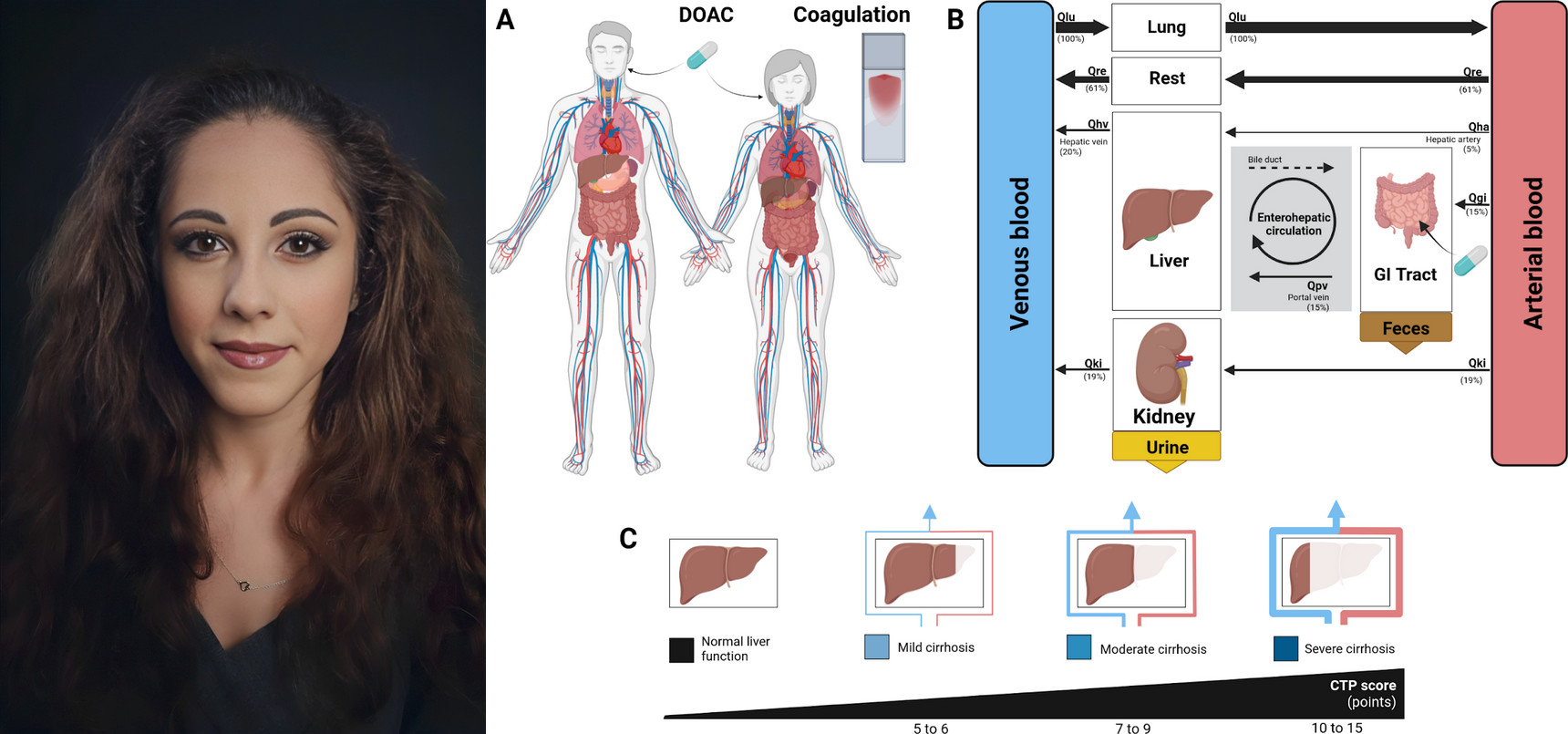
Launch of New Research Internship in PBPK/PD Modeling
|
PBPK Model Development for Tirzepatide in Diabetes and Obesity Treatment We are pleased to announce that Elisabetta Cassabianca will be joining the König Lab to work on an exciting new project focused on developing a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of the direct oral anticoagulant (DOAC) rivaroxaban. Since their introduction, DOACs have revolutionized anticoagulant therapy, offering significant advantages over traditional vitamin K antagonist therapy, such as reduced dietary restrictions and less frequent monitoring. However, despite their widespread adoption, there remain gaps in understanding their long-term efficacy, safety, and impact on specific patient populations. Elisabetta’s project aims to address these knowledge gaps by constructing a comprehensive PBPK/PD model of rivaroxaban. This computational model will serve as a digital twin of the human body, enabling precise predictions of the drug’s pharmacokinetics (absorption, distribution, metabolism, and excretion) and pharmacodynamics (anticoagulant effects). The model will be instrumental in exploring the influence of various factors, such as age, gender, hepatic or renal impairment, and genetic variations, on rivaroxaban therapy. |
2025-01-07
Launch of New Research Internship in PBPK/PD Modeling

|
PBPK Model Development for Tirzepatide in Diabetes and Obesity Treatment Abhinav Mishra, a Master Intern, will develop a Physiologically Based Pharmacokinetic (PBPK) model for tirzepatide (Mounjaro, Zepbound), a dual agonist for GLP-1 and GIP receptors used in treating type 2 diabetes mellitus (T2DM) and obesity. The project will focus on analyzing the drug’s pharmacokinetics (PK) and pharmacodynamics (PD), with particular attention to the effects of renal and hepatic impairments. By building a computational model and integrating patient-specific data, the research aims to better understand factors influencing individual responses and improve the clinical use of tirzepatide in diverse patient populations. |
2024-12-20
Circle U. Funds AlgoNomy to Combat Digital Paternalism in AI-Driven Healthcare

|
Circle U. has funded AlgoNomy: Advancing Doctor-Patient Autonomy in AI-Driven Healthcare, a collaborative project tackling the issue of digital paternalism—where algorithms dominate healthcare decisions over human collaboration. Partnering with six leading European universities, AlgoNomy aims to develop ethical, transparent, and patient-centered AI systems that preserve autonomy and promote shared decision-making. https://www.circle-u.eu/resources/seed-funded-projects/ The Circle U. European University Alliance has announced funding for AlgoNomy: Advancing Doctor-Patient Autonomy in AI-Driven Healthcare, a pioneering project aimed at tackling the emerging issue of digital paternalism—an adaptation of medical paternalism where healthcare decisions are increasingly driven by algorithms rather than collaborative input from patients and physicians. Led by a consortium of six leading universities—UniPi (University of Pisa), KCL (King's College London), PC (Université Paris Cité), BU (University of Belgrade), UW (University of Vienna), and HU (Humboldt Universität zu Berlin)—AlgoNomy will focus on developing ethical, technical, and practical solutions to ensure AI empowers rather than replaces human autonomy in clinical decision-making. Funded under the Circle U. initiative, AlgoNomy is dedicated to fostering transparent, patient-centered, and clinician-friendly AI systems that promote shared decision-making and responsible innovation in healthcare. This ambitious project marks a significant step in redefining the role of AI in medicine, ensuring technology serves as a tool for collaboration, not control. Stay tuned for updates on how AlgoNomy is shaping the future of ethical AI in healthcare! |
2024-10-01
Humboldt Internship Program 2025 - Computational Modeling of Drug Detoxification - A Systems Medicine Approach

|
We offer Internships in 2025, academic three-month research stays at Humboldt-Universität zu Berlin. The application for the Summer Term Internships 2025 is open from 11 November to 20 December 2024. https://hic.hu-berlin.de/en/internship-program/projects/201
Humboldt Internship Program is an international short-term program for subject-specific, experiential learning. It allows participants to work with teams in research projects and university-related start-ups for three months. |
2024-08-30
Launch of Two New Research Projects in PBPK/PD Modeling
|
Ennie Tensil and Michelle Elias start Bachelor projects on losartan and glimeperide We are excited to announce the initiation of two projects within our group, focusing on the development of Physiologically-Based Pharmacokinetic/Pharmacodynamic (PBPK/PD) models. These projects aim to deepen our understanding of drug action and variability in treatment responses, contributing significantly to personalized medicine. Michelle Elias will lead the first project, which centers on the development of a PBPK/PD model for the sulfonylurea glimepiride, a widely used medication for managing type 2 diabetes. This project aims to enhance our understanding of the intraindividual variability observed in diabetes treatment with sulfonylureas, particularly focusing on differences in hepatorenal function. The insights gained from this model could lead to more personalized and effective treatment strategies for patients with diabetes. The second project, led by Ennie Tensil, will focus on developing a PBPK/PD model for the angiotensin II receptor blocker (ARB) losartan. This medication is commonly prescribed for treating high blood pressure and protecting against stroke in high-risk individuals. The project will investigate the physiological factors influencing the pharmacokinetics and pharmacodynamics of losartan, aiming to improve our understanding of its efficacy and variability in different patient populations. Both projects represent important advancements in our research capabilities, with the potential to impact clinical practice by providing deeper insights into drug behavior and patient variability. We look forward to the outcomes these studies will bring and the contributions they will make to the field of pharmacology. |
2024-08-28
Master Thesis Jonas Küttner
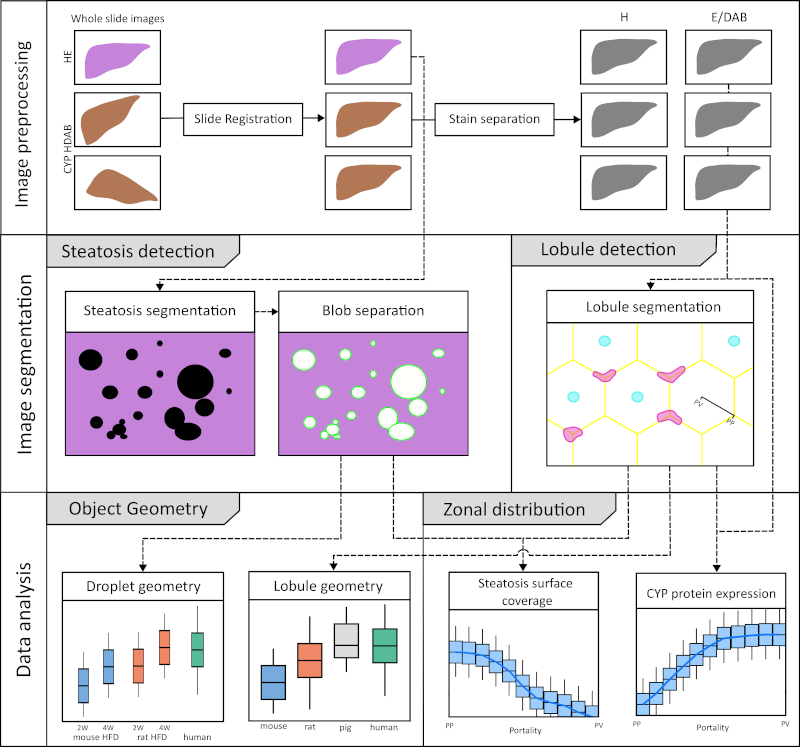
|
Jonas Küttner submitted his Master thesis developing a workflow for image analysis in the liver /paper/theses/Master.Thesis.Jonas.Kuettner.pdf The mammalian liver is organized into three-dimensional structures called lobules that are integral to its function. According to the widely accepted hexagonal model, the cross-section of each lobule is characterized by a central vein and portal triads at the corners. These portal triads consist of a hepatic portal vein, a hepatic arteriole, and a bile duct. Blood is delivered through the hepatic artery and portal vein and flows inward from the outer periportal region toward the central vein through sinusoids lined with hepatocytes. These hepatocytes express a wide variety of metabolic enzymes, including cytochrome P450 (CYP) isoforms that are critical for xenobiotic metabolism. Notably, many hepatic enzymes, including CYPs, exhibit differential expression along the periportal-to-perivenous axis, a spatial variation known as liver zonation. Non-alcoholic fatty liver disease (NAFLD) is one of the most common liver diseases characterized by the accumulation of fat in the liver (steatosis) in the absence of significant alcohol consumption. Histopathologically, NAFLD is characterized by the presence of microvesicular fat droplets within hepatocytes. Although liver zonation has been extensively studied, the protein gradients of CYPs and their alterations in response to steatosis have not been systematically quantified or compared across species. This study aimed to develop an image analysis workflow to accurately derive these lobular expression gradients and quantify macrosteatosis in whole slide images (WSI) of liver histology. A comprehensive analysis was performed on whole slide images (WSIs) of mouse, rat, pig, and human liver specimens. These images included hematoxylin and eosin (H&E) stained slides and immunohistochemically stained slides for glutamine synthetase (GS) and cytochrome P450 isoforms 1A2, 2E1, 2D6, and 3A4. To achieve the study objectives, two image analysis workflows were developed: one using classical methods and the other using convolutional neural networks (CNN). These workflows were designed to perform two key tasks: (1) segmentation of liver lobules and (2) detection of macrosteatosis droplets. Key findings were that there was significant variability in lobular geometry within subjects, with strong consistency across species, with inter-species differences observed primarily in lobular size. A slight increase in lobular size was observed in steatotic samples, with a moderate to strong correlation between lobular size and steatosis content. In addition, interspecies differences in zonation patterns were observed for CYP1A2 in pigs, CYP2E1 in rats, and CYP3A4 in humans. Importantly, zonation patterns in steatotic subjects were not qualitatively different from those in non-steatotic subjects across species. In summary, I have successfully developed and implemented an image analysis workflow for whole slide images (WSIs) that allows detailed examination of liver lobule geometry and macrosteatosis droplets. Furthermore, the resulting datasets were integrated to facilitate a comprehensive analysis of the zonal distribution of CYP protein expression and macrosteatosis within the lobules. |
2024-08-08
Open Science Ambassador of the BUA

|
Proud to be selected as Open Science Ambassador of the Berlin University Alliance (BUA) in 2024. https://www.berlin-university-alliance.de/en/commitments/research-quality/open-science/index.html To find discipline-specific and accessible solutions to be implemented into everyday research routines, we have applied for Open Science Ambassador. As part of the work we will work on the reproducibility of computational models. |
2024-07-18
Participation in DHPE

|
Matthias König selected for the Digital Health Professions Educator (DHPE) Program https://dsfz.charite.de/hochschuldidaktik/digital_health_professions_educator/
We are excited to announce that Matthias König has been selected to participate in the "Digital Health Professions Educator" (DHPE) program for 2024/25. This program, part of the "HEDS: Handlungs- und Entscheidungskompetenz Digital Stärken" project, aims to enhance digital teaching skills at Charité. Running from June 2024 to June 2025, the program includes monthly in-person sessions and digital self-study units, leading to a nationally recognized certification (MQ II). |
2024-07-18
Humboldt Internship Program (HIP) conference

|
Proud to participate in Humboldt Internship Program 2024. Great work by Vera, Deepa and Prerna. https://hic.hu-berlin.de/en/internship-program Students presented their results during the Humboldt Internship Program 2024. Great work by Vera, Deepa and Prerna. |
2024-07-15
Final ecosystem meeting of EDITH on building the VHT

|
Proud to participate in the final ecosystem meeting of EDITH on building the virtual human twin (VHT) in Amsterdam. The first day was dedicated to a plenary session on the EDITH proof of concept infrastructure, presented from the users’ perspective, where Researchers, Communities, together with Healthcare Providers and Patients were well represented to connect resources and enlighten the federated nature of the VHT, as well as to deploy VHT at bed side in clinical workflows and allow patients to upload their own data in the VHT, while representatives from Industry explored possible interaction of services within VHT. |
2024-07-01
New project: Vera Tereshchuk starts SGLT2 PBPK Modeling Project
|
Vera Tereshchuk will develop a PBPK models for canagliflozin, aiming to enhance understanding of pharmacokinetics and pharmacodynamics in diabetes. Vera Tereshchuk is developing a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of the SGLT2 inhibitor canagliflozin. The objective is to enhance understanding of intraindividual variability in diabetes treatment with SGLT2 inhibitors, focusing on differences in hepatorenal function. |
2024-06-25
PAGE2024: Presentation of our research in Rome

|
Mariia Myshkina and Matthias König presented our captopril modeling results during the PAGE2024 (Population Approach Group Europe) meeting in Rome. Introduction: Angiotensin-converting enzyme (ACE) inhibitors, such as captopril, are widely prescribed for the treatment of hypertension and heart failure. Captopril, the first ACE inhibitor developed in the 1980s, is primarily taken orally and is rapidly absorbed, reaching peak plasma concentrations within an hour. Its half-life is approximately 2 hours, but several factors, including renal function, heart disease, age and sex, can affect its pharmacokinetics. Despite the widespread use of captopril, comprehensive pharmacokinetic data have been lacking. Our group has developed PK-DB (https://pk-db.com) [1], an open database containing high-quality pharmacokinetic data from clinical and preclinical research. PK-DB includes patient cohort characteristics, interventions, concentration-time profiles, and kinetic parameters, with the ability to automatically calculate pharmacokinetic parameters using non-compartmental methods. Objectives: The objectives of this study were to curate and analyze captopril pharmacokinetic data, expand the PK-DB, develop a whole-body physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of captopril in accordance with FAIR principles [2], and improve understanding of the intra-individual variability in captopril pharmacokinetics influenced by factors such as age, sex, renal and cardiac function. Methods: Over 600 publications on captopril pharmacokinetics were reviewed through a systematic literature search, with a subset selected for data curation in PK-DB. The PBPK model was developed using a compartmental approach and encoded in Systems Biology Markup Language (SBML) [3] for accessibility and reproducibility. Results: A comprehensive set of captopril pharmacokinetic data was generated from 15 clinical studies in healthy volunteers and patients with renal impairment, heart failure and hypertension. The PBPK model includes four major compartments (blood, intestine, liver, kidney) and describes absorption, distribution, metabolism and excretion. It successfully represents the pharmacokinetic profile of captopril in different age groups, showing minimal age-related differences in unchanged drug concentration. Heart failure did not significantly affect maximum plasma concentration or area under the curve (AUC), but renal function strongly influenced captopril elimination. We present this data set and open PBPK model of captopril as a valuable resource for further research. Conclusions: This study successfully establishes a freely accessible, comprehensive dataset and an open PBPK model of captopril, enhancing our understanding of its pharmacokinetic behavior across different patient groups. Our findings emphasize the significance of renal function in captopril elimination and provide a valuable resource for personalized medicine approaches in hypertension and heart failure treatment. |
2024-06-09
Bachelor Thesis Afruja Hossain
|
Afruja Hossain finished her Bachelor thesis developing a database of CYP and UGT enzymes /paper/theses/Bachelor.Thesis.Afruja.Hossain.pdf Afruja Hossain has successfully finished her Bachelor thesis titled 'A Systematic Overview of Protein Variability in Cytochrome P450 and UDP-glucuronosyltransferase Enzymes in the Human Liver.' Her research provides an in-depth analysis of the key liver enzymes Cytochrome P450 (CYP450) and UDP-glucuronosyltransferase (UGT), crucial for drug metabolism. The study highlights the variability in protein levels of these enzymes, influenced by factors such as age, smoking, and alcohol consumption. Key findings include: - Creation of a database of CYP and UGT protein levels in the human liver. - Identification of significant individual variability in enzyme protein levels. - Correlations between different CYP and UGT isoforms. Afruja's work offers valuable insights into drug metabolism and could enhance understanding of drug interactions and toxicity. Congratulations to Afruja on her significant academic achievement. |
2024-06-07
Successful Completion of One-Week Pharmacokinetic Modelling Course
|
We are excited to announce the successful completion of our one-week intensive course on Pharmacokinetic Modelling, where students delved into principles, drug distribution, clearance, elimination, and various modelling techniques to enhance drug therapy and development. Pharmacokinetic modelling is the study of how drugs are absorbed, distributed, metabolised and excreted in the body. The pharmacokinetic modelling course covers topics such as pharmacokinetic principles, drug distribution, clearance and elimination and the factors that influence these processes. Students will learn about different models used to describe pharmacokinetics, such as compartmental models and physiologically based pharmacokinetic models, and how these models can be used to predict drug concentrations and optimise dosing regimens. Other topics that may be covered include pharmacodynamics, drug-drug interactions and the use of pharmacokinetic modelling in drug development and clinical practice. Overall, a course in pharmacokinetic modelling will provide students with a comprehensive understanding of the principles and techniques used to describe the movement of drugs through the body and how this knowledge can be applied to improve drug therapy. |
2024-05-23
New Project Launch via Humboldt Internship Program (HIP): PK-LLM by Prerna Parakkat
|
We are excited to announce the start of a new project under the Humboldt Internship Program (HIP) by Prerna Parakkat. Prerna will be working on PK-LLM: Implementing a Large Language Model (LLM) as an Expert System for Pharmacokinetic (PK) Data Curation from Scientific Publications. The project focuses on creating a specialized LLM tailored to interpret and extract pharmacokinetic data from diverse scientific texts, automating the extraction of key data such as parameters, dosing information, and subject details. It also aims to integrate this system with existing pharmacokinetic databases and computational modeling workflows for digital twins. We anticipate significant improvements in the accuracy and efficiency of pharmacokinetic data extraction and curation, setting a new standard in the field. |
2024-05-16
Preprint: Cross-Species Variability in Lobular Geometry and Cytochrome P450 Hepatic Zonation: Insights into CYP1A2, CYP2E1, CYP2D6 and CYP3A4
|
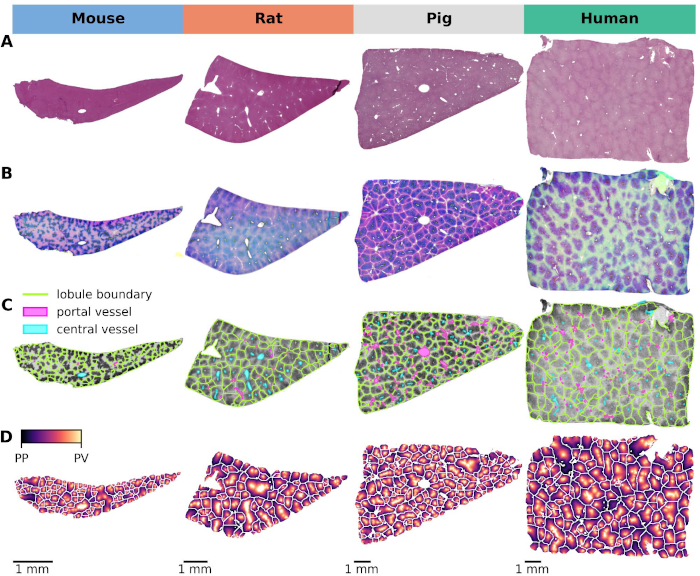
Our latest publication on 'Cross-Species Variability in Lobular Geometry & Cytochrome P450 Hepatic Zonation' reveals insights into liver function across species using WSI technology. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2024.1404938/full This study explores the critical interplay between lobular geometry and the zonated distribution of cytochrome P450 (CYP) enzymes across species. We present an innovative approach to assess lobular geometry and zonation patterns using whole slide imaging (WSI). This method allows a detailed, systematic comparison of lobular structures and spatial distribution of key CYP450 enzymes and glutamine synthetase in four different species (mouse, rat, pig, and human). Our results shed light on species differences in lobular geometry and enzymatic zonation, providing critical insights for drug metabolism research. Based on our approach we could determine the minimum number of lobules required for a statistically representative analysis, an important piece of information when evaluating liver biopsies and deriving information from WSI. |
2024-04-15
New projects: Nike Nemitz and Yusuf Ali Kulanoglu start PBPK Modeling Projects
|
Nike Nemitz and Yusuf Ali Kulanoglu will develop PBPK models for dapagliflozin and aliskiren, respectively, aiming to enhance understanding of pharmacokinetics and pharmacodynamics in diabetes and cardiovascular treatments. Nike Nemitz is developing a physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) model of the SGLT2 inhibitor dapagliflozin. The objective is to enhance understanding of intraindividual variability in diabetes treatment with SGLT2 inhibitors, focusing on differences in hepatorenal function. Yusuf Ali Kulanoglu is working on a PBPK model of the direct renin inhibitor aliskiren to improve understanding of its pharmacokinetics and the influencing factors. Both projects aim to provide deeper insights into drug behavior and variability, potentially improving therapeutic outcomes. |
2024-04-15
Publication: The Simulation Experiment Description Markup Language (SED-ML): Language Specification for Level 1 Version 5

|
Level 1 Version 5 of SED-ML enhances the ability to annotate, archive, share, and reproduce computational simulation experiments by allowing modelers to define tasks, model changes, ranges, and outputs using the Kinetic Simulation Algorithm Ontology (KiSAO). https://www.degruyter.com/document/doi/10.1515/jib-2024-0008/html We are excited to announce the latest publication titled "The Simulation Experiment Description Markup Language (SED-ML): Language Specification for Level 1 Version 5." This publication addresses the growing need in modern biological research for methods to annotate, archive, share, and reproduce computational simulation experiments. As computational simulations become more integral to biological research, extensive collaboration among modelers, experimentalists, and engineers is required. The Minimum Information About a Simulation Experiment (MIASE) guidelines provide a framework for sharing these experiments, and SED-ML offers a computer-readable format for the information outlined by MIASE. Level 1 Version 5 of SED-ML significantly enhances the capabilities for modelers by allowing the definition of tasks, model changes, ranges, and outputs using the Kinetic Simulation Algorithm Ontology (KiSAO). This builds on Version 4, which permitted defining simulations entirely with KiSAO. SED-ML growing ecosystem includes a wide range of investigators, model languages, and software tools, supporting various simulation tools, visual editors, model repositories, and validators. For more information about SED-ML and its applications, visit https://sed-ml.org. |
2024-05-05
Master Thesis Shubhankar Palwankar
|
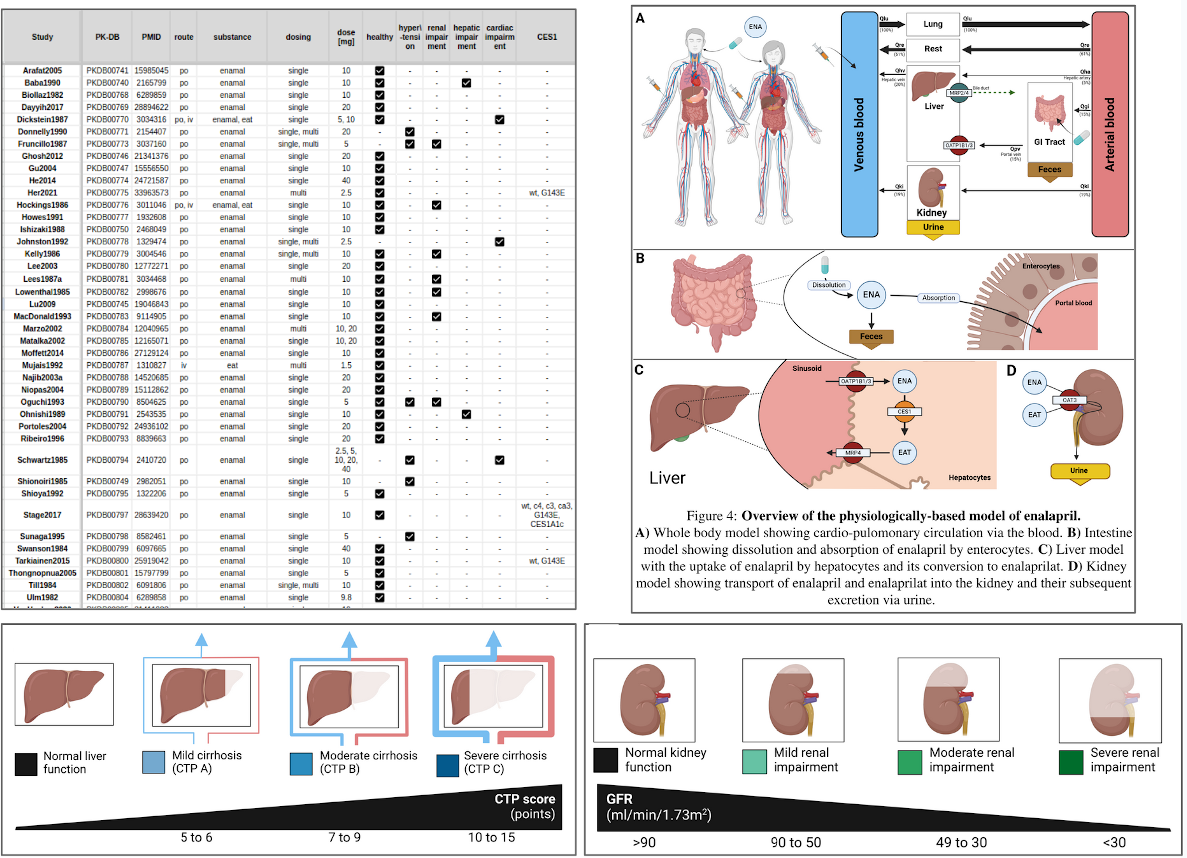
Shubhankar Palwankar defended his Master Thesis developing a PBPK model of enalapril /paper/theses/Master.Thesis.Shubhankar.Palwankar.pdf We are pleased to announce that Shubhankar Palwankar has successfully defended his Master Thesis, which focuses on the development of a Physiologically Based Pharmacokinetic (PBPK) model of enalapril, a key medication used to treat high blood pressure and other cardiovascular conditions. Enalapril works as an ACE inhibitor, playing a crucial role in regulating blood pressure through the renin-angiotensin-aldosterone system (RAAS). Shubhankar's research involved the creation of an extensive database from 49 clinical trials, which was used to parameterize and validate his computational model. His model effectively simulates the pharmacokinetics of enalapril and its active metabolite, enalaprilat, under various conditions including renal and hepatic impairment and changes in CES1 enzyme activity. The findings from this study offer valuable insights into how different physiological impairments affect the drug's behavior in the body, showing excellent agreement with clinical data. The developed model and data are publicly accessible in SBML format under a CC-BY 4.0 license from the PK-DB pharmacokinetics database, contributing to open science and further research in pharmacokinetics. Congratulations to Shubhankar Palwankar for his significant contributions to pharmacological research and his successful thesis defense. |
2024-03-29
New multiscale liver model unveils zonation-function dynamics in Drug-Induced Liver Injury (DILI).

|
We're excited to announce the preprint of our latest research, 'Simulation of zonation-function relationships in the liver using coupled multiscale models: Application to drug-induced liver injury.' This study introduces a multiscale digital twin of the liver, integrating models from cellular to lobular scales, and sheds light on how liver zonation patterns influence drug-induced liver injury. https://doi.org/10.1101/2024.03.26.586870 Multiscale modeling requires the coupling of models on different scales, often based on different mathematical approaches and developed by different research teams. This poses many challenges, such as defining interfaces for coupling, reproducible exchange of submodels, efficient simulation of the models, or reproducibility of results. Here, we present a multiscale digital twin of the liver that couples a partial differential equation (PDE)-based porous media approach for the hepatic lobule with cellular-scale ordinary differential equation (ODE)-based models. The models based on the theory of porous media describe transport, tissue mechanical properties, and deformations at the lobular scale, while the cellular models describe hepatic metabolism in terms of drug metabolism and damage in terms of necrosis. The resulting multiscale model of the liver was used to simulate perfusion-zonation-function relationships in the liver spanning scales from single cell to the lobulus. The model was applied to study the effects of (i) protein zonation patterns (metabolic zonation) and (ii) drug concentration dependence on spatially heterogeneous liver damage in the form of necrosis. Depending on the zonation pattern, different liver damage patterns could be reproduced, including periportal and pericentral necrosis as seen in drug-induced liver injury (DILI). Increasing the drug concentration led to an increase in the observed damage pattern. A key point for the success was the integration of domain-specific simulators based on standard exchange formats, i.e., libroadrunner for the high-performance simulation of ODE-based systems and FEBio for the simulation of the continuum-biomechanical part. This allows a standardized and reproducible exchange of cellular scale models in the Systems Biology Markup Language (SBML) between research groups. |
2024-03-01
PBPK Model of Morphine Project
|
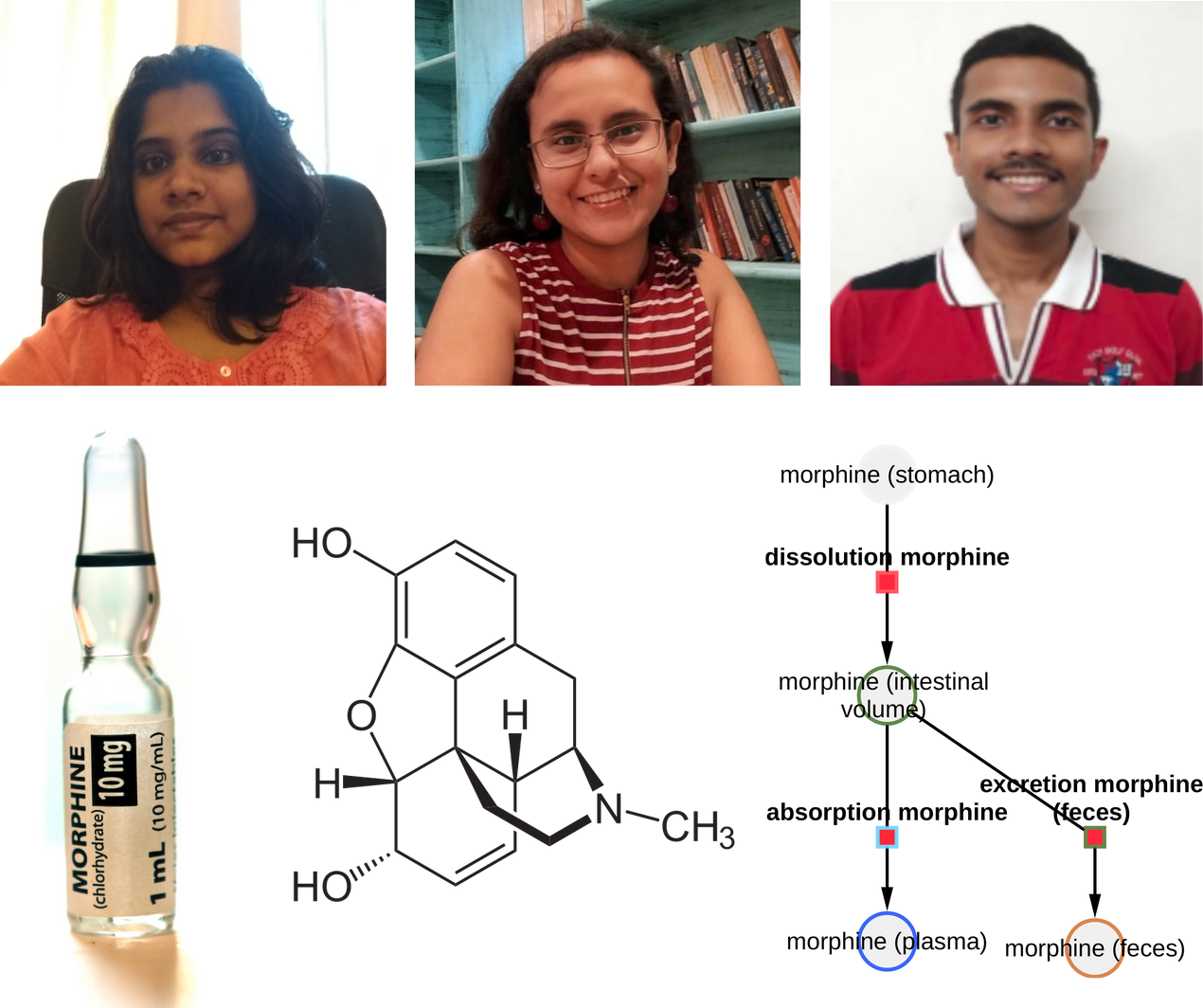
New Internship Project Launch: Development of a Physiologically Based Pharmacokinetics Model of Morphine. Deepa supported by Rohini and Rohit will develop a PBPK model of morphine. Deepa supported by Rohini and Rohit will develop a physiologically based pharmacokinetic (PBPK) model of morphine. Morphine, a potent analgesic derived from the opium poppy, is essential for pain management but poses significant pharmacokinetic challenges. This project aims to address intra-individual variability in morphine treatment by establishing a comprehensive pharmacokinetics/pharmacodynamics database and constructing a whole-body computational model. Key areas of focus include the role of the liver and kidney in morphine metabolism and excretion, the effects of renal and hepatic impairment, and the effects of different morphine formulations and delivery methods. This research aims to improve the safe and effective use of morphine in clinical settings, taking into account factors such as age and genetic polymorphisms. |
2024-02-01
Mariia Myshkina has started her PhD in the ATLAS project.

|
We welcome Mariia to our team who will be working on computational modeling of the liver within ATLAS. Mariia will develop PBPK models of the liver in HCC coupled with machine learning approaches. |
2024-01-18
Participation in the First EDITH-CSA Ecosystem Meeting - Advancing the Virtual Human Twin

|
We participated in the first EDITH-CSA 'Ecosystem meeting on building the Virtual Human Twin was held at the École Polytechnique de Paris on January 18 and 19, 2024. After the official launch of the Virtual Human Twin initiative and the VHT Manifesto in December, the first open community meeting attracted over 180 registered guests from the EDITH-CSA consortium and the wider ecosystem. The enthusiasm of the wider community to travel to Paris (amidst difficult weather conditions) and proactively contributing towards developing the roadmap for the VHT was inspiring. |
2023-12-29
Preprint: Cross-Species Variability in Lobular Geometry and Cytochrome P450 Hepatic Zonation: Insights into CYP1A2, CYP2E1, CYP2D6 and CYP3A4

|
Our latest preprint on 'Cross-Species Variability in Lobular Geometry & Cytochrome P450 Hepatic Zonation' reveals insights into liver function across species using WSI technology. https://www.biorxiv.org/content/10.1101/2023.12.28.573567v1 This study explores the critical interplay between lobular geometry and the zonated distribution of cytochrome P450 (CYP) enzymes across species. We present an innovative approach to assess lobular geometry and zonation patterns using whole slide imaging (WSI). This method allows a detailed, systematic comparison of lobular structures and spatial distribution of key CYP450 enzymes and glutamine synthetase in four different species (mouse, rat, pig, and human). Our results shed light on species differences in lobular geometry and enzymatic zonation, providing critical insights for drug metabolism research. Based on our approach we could determine the minimum number of lobules required for a statistically representative analysis, an important piece of information when evaluating liver biopsies and deriving information from WSI. |
2023-11-03
Eleven Strategies for Making Reproducible Research and Open Science Training the Norm at Research Institutions

|
We are pleased to announce our latest paper in eLife that provides a roadmap for integrating reproducible research and open science practices into academic work. https://elifesciences.org/articles/89736 We are excited to announce that our recent paper, "Eleven Strategies for Making Reproducible Research and Open Science Training the Norm at Research Institutions," has been published in eLife, offering a roadmap for integrating reproducible research and open science practices into academic work. Developed in collaboration with the German Reproducibility Network, the paper proposes eleven strategies focusing on modifying research assessment criteria, enhancing training, and fostering community support. This work aims to encourage researchers and institutions to adopt practices that increase the reliability and accessibility of scientific research. |
2023-11-03
Your Dietary Digitial Twin (DDtwin)

|
Advancing Precision Nutrition: Workshop Success on Dietary Digital Twin, a workshop in Leiden from 30 October - 3 November 2023. https://www.lorentzcenter.nl/your-dietary-digital-twin-ddtwin.html
We are excited to announce our participation in a groundbreaking workshop dedicated to the development of the Dietary Digital Twin (DDtwin) technology platform. This innovative endeavor aims to provide precision nutrition by integrating biology-based and data-driven models with real-life data, harnessing the power of wearable sensors and applications. |
2023-10-31
Humboldt Internship Program 2024 - Computational Modeling of Drug Detoxification - A Systems Medicine Approach

|
We offer Internships in 2024, academic three-month research stays at Humboldt-Universität zu Berlin. The application for the Summer Term Internships 2024 is open from 03 November to 10 December 2023. https://hic.hu-berlin.de/en/internship-program/projects/201
Humboldt Internship Program is an international short-term program for subject-specific, experiential learning. It allows participants to work with teams in research projects and university-related start-ups for three months. |
2023-10-10
e:Med Meeting 2023 on Systems Medicine: Quantifying Fat Zonation in Liver Lobules: An Integrated Multiscale In-silico Model

|
We successfully presented our research on reproducible digital twins for personalized liver function assessment at the e:Med Meeting 2023 on Systems Medicine. https://www.youtube.com/watch?v=6faFifoXUeo
Essential prerequisites for the practical application and translation of computational models include: i) reproducibility of results; ii) model reusability and extensibility; iii) data availability; and iv) strategies for model stratification and individualization. Here, we present a modeling workflow built around these foundational prerequisites, with a focus on liver function tests. |
2023-09-18
Preprint: Quantifying Fat Zonation in Liver Lobules: An Integrated Multiscale In-silico Model
|
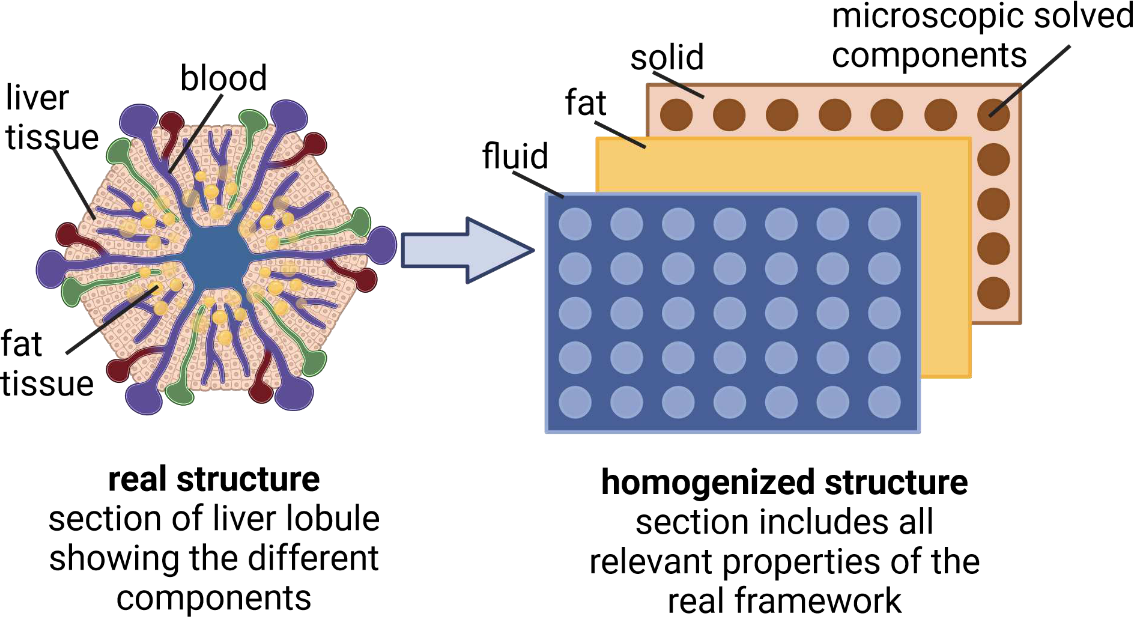
Our latest preprint is now out: An integrated multiscale model for quantifying fat zonation in liver lobules. https://doi.org/10.21203/rs.3.rs-3348101/v1 We have developed a sophisticated computer model to explore how various factors contribute to the distribution and accumulation of fat in the liver, a phenomenon known as "metabolic zonation." This model will help in understanding how liver health is affected by different conditions and could play a crucial role in studying diseases like MASLD (metabolic dysfunction-associated steatotic liver disease). By simulating liver functions, the model considers interactions between blood flow, oxygen levels, and fat metabolism within liver lobules. |
2023-08-28
Hannah Menghis from Brown University starts her internship

|
We welcome Hannah Menghis who will establish a PBPK model of semaglutide. Hannah will establish a pharmacokinetic dataset of semaglutide and develop an initial version of a physiologically based pharmacokinetic (PBPK) model of the GLP-1 inhibitor semaglutide. The objective is to enhance our understanding of the underlying causes of intraindividual variability in semaglutide treatment, e.g., differences in renal function. |
2023-08-02
A pathway model of glucose-stimulated insulin secretion in the pancreatic β-cell
|
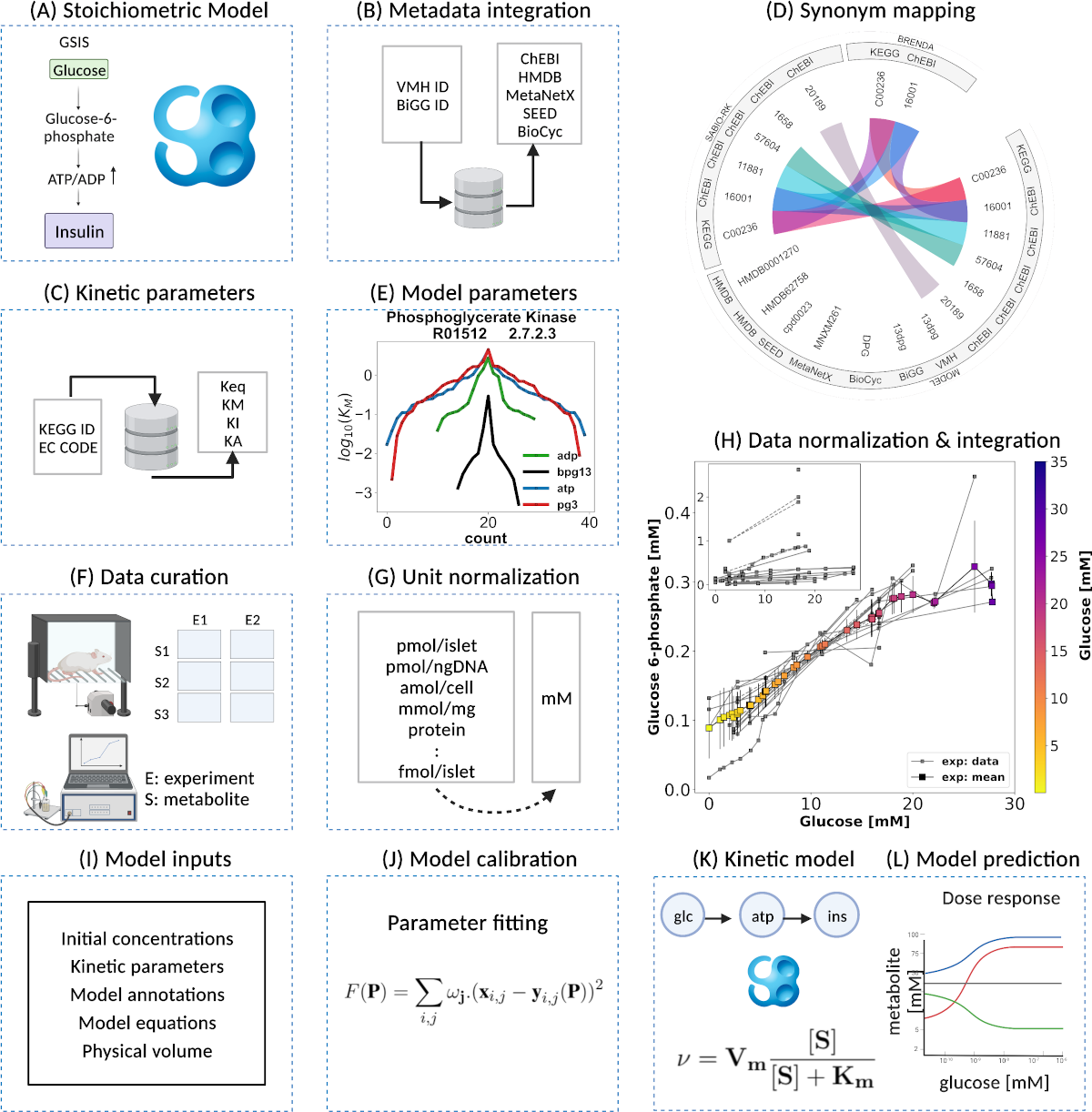
Our latest publication is now on Frontiers of Endocrinology: A pathway model of glucose-stimulated insulin secretion in the pancreatic β-cell https://doi.org/10.3389/fendo.2023.1185656 The pancreas plays a critical role in maintaining glucose homeostasis through the secretion of hormones from the islets of Langerhans. Glucose-stimulated insulin secretion (GSIS) by the pancreatic β-cell is the main mechanism for reducing elevated plasma glucose. Here we present a systematic modeling workflow for the development of kinetic pathway models using the Systems Biology Markup Language (SBML). Steps include retrieval of information from databases, curation of experimental and clinical data for model calibration and validation, integration of heterogeneous data including absolute and relative measurements, unit normalization, data normalization, and model annotation. An important factor was the reproducibility and exchangeability of the model, which allowed the use of various existing tools. The workflow was applied to construct a novel data-driven kinetic model of GSIS in the pancreatic β-cell based on experimental and clinical data from 39 studies spanning 50 years of pancreatic, islet, and β-cell research in humans, rats, mice, and cell lines. The model consists of detailed glycolysis and phenomenological equations for insulin secretion coupled to cellular energy state, ATP dynamics and (ATP/ADP ratio). Key findings of our work are that in GSIS there is a glucose-dependent increase in almost all intermediates of glycolysis. This increase in glycolytic metabolites is accompanied by an increase in energy metabolites, especially ATP and NADH. One of the few decreasing metabolites is ADP, which, in combination with the increase in ATP, results in a large increase in ATP/ADP ratios in the β-cell with increasing glucose. Insulin secretion is dependent on ATP/ADP, resulting in glucose-stimulated insulin secretion. The observed glucose-dependent increase in glycolytic intermediates and the resulting change in ATP/ADP ratios and insulin secretion is a robust phenomenon observed across data sets, experimental systems and species. Model predictions of the glucose-dependent response of glycolytic intermediates and biphasic insulin secretion are in good agreement with experimental measurements. Our model predicts that factors affecting ATP consumption, ATP formation, hexokinase, phosphofructokinase, and ATP/ADP-dependent insulin secretion have a major effect on GSIS. In conclusion, we have developed and applied a systematic modeling workflow for pathway models that allowed us to gain insight into key mechanisms in GSIS in the pancreatic β-cell. |
2023-07-17
Bachelor Thesis Beatrice Stemmer Mallol
|
Beatrice Stemmer Mallol submitted her Bachelor thesis developing a PBPK model of talinolol /paper/theses/Bachelor.Thesis.Beatrice.Stemmer.Mallol.pdf Congratulations to Bea for successfully submitting her Bachelor thesis on a physiologically based pharmacokinetic (PBPK) model of the probe drug talinolol for the characterization of intestinal P-glycoprotein. Talinolol, a cardioselective beta-blocker applied in treating various cardiovascular diseases and tachyarrhythmias, is widely adopted as a probe drug for the intestinal efflux transporter P-glycoprotein. In the human body, P-gp plays a crucial role, given its distribution across various tissues to protect against potentially toxic substances, while expediting the elimination of xenobiotics. By employing talinolol for P-gp phenotyping, researchers can evaluate factors that influence P-gp-mediated transport, such as P-gp genetic polymorphisms and its distribution throughout the intestine. Bea's thesis presents a comprehensive dataset on talinolol pharmacokinetics, the study of how the body absorbs, distributes, metabolizes, and excretes drugs. This data was used to develop a PBPK model for talinolol, offering detailed insights into its behaviour within the body. The new model investigates the impact of numerous factors on talinolol's pharmacokinetics, including the genetic variants of P-gp, enzymatic activity of transporters OATP2B1 and OATP1B1, and site-specific distribution of P-gp and OATP2B1 proteins in the intestine. It also investigates the impact of diseases like liver cirrhosis and renal dysfunction. |
2023-07-07
PhD Defense of Jan Grzegorzewski
|
Jan Grzegorzewski defended his PhD thesis on physiologically based pharmacokinetic modeling (PBPK) for dynamical liver function tests and Cytochrome P450 (CYP) phenotyping. /paper/presentations/2021-07-07_Defense.Jan.Grzegorzewski Congratulations to Jan Grzegorzewski for successfully defending his PhD thesis on physiologically based pharmacokinetic modeling (PBPK) for dynamical liver function tests and Cytochrome P450 (CYP) phenotyping. Jan developed the extensive open pharmacokinetics database, PK-DB, which helped to demonstrate demographic biases and errors in existing pharmacokinetic literature. His research includes a meta-analysis on caffeine pharmacokinetics, discovering multiple factors influencing liver function and CYP1A2 activity. Furthermore, he developed a PBPK model to predict individual plasma concentrations of dextromethorphan based on the CYP2D6 genotype and physiological traits. His work is expected to significantly enhance the precision of dynamic liver function tests and advance personalized medicine. |
2023-07-01
Xenia Petukhova and Zahra Bahri start Humboldt Internship Program

|
Humboldt Internship Program 2023 - Computational Modeling of Drug Detoxification - A Systems Medicine Approach We welcome Xenia and Zhara in our group for the Humboldt Internship Program 2023. Xenia and Zhara will develop a physiologically based pharmacokinetic (PBPK) model for inulin and creatinine to assess the glomerular filtration rate (GFR). Our goal is to gain a deeper understanding of the impact of various inulin protocols on GFR measurement and the influence of physiological factors, such as body composition, on GFR determination using inulin. Furthermore, it should enhance our understanding of how variations in creatinine production, muscle mass, and tubular secretion of creatinine influence eGFR calculations. |
2023-06-16
Keynote: Advancing Liver Function Assessment: Personalized and Stratified Approaches with Standardized Computational Models and Data

|
Keynote talk at the Workshop on Computational Models in Biology and Medicine 2023 organized by Nicole Radde and Sebastian Höpfl. https://docs.google.com/presentation/d/1hi7-4bwzqFJYiYQkbSBHNQivSPA5T3YHB9OLZqJG0rA/edit?usp=sharing Essential prerequisites for the practical application and translation of computational models include: i) reproducibility of results; ii) reusability and extensibility of models; iii) data availability; and iv) strategies for model stratification and individualization. In this study, we present a modeling workflow tailored to these critical aspects, with a focus on liver function tests. Evaluating liver function is a crucial task in hepatology, yet accurately quantifying hepatic function has persisted as a clinical challenge. Dynamic liver function tests offer a promising method for non-invasive in vivo assessment of liver function and metabolic phenotyping. These clinical tests determine liver function through the elimination of a specific test substance, thus revealing information about the liver's metabolic capacity. We employed whole-body physiologically-based pharmacokinetic (PBPK) models to simulate these tests, which encompass absorption, distribution, metabolism, and elimination processes. PBPK models serve as powerful instruments for investigating drug metabolism and its impact on the human body. In this research, we showcase our efforts in utilizing PBPK models as digital twins for metabolic phenotyping and liver function evaluation. To develop and validate our models, we created the first open pharmacokinetics database, PK-DB, containing curated data from over 600 clinical studies. Our models are individualizable and stratifiable, enabling simulation of lifestyle factors and co-administration effects on drug metabolism. We have applied our models to various clinical inquiries, such as simulating individual outcomes post-hepatectomy using an indocyanine green model and examining the influence of CYP2D6 gene variants through a dextromethorphan model integrated with drug-gene interactions. These models are constructed hierarchically, describing metabolic and other biological processes in organs like the liver and kidneys, connected to whole-body physiology. All models and data are accessible for reuse in a reproducible manner, encoded in the Systems Biology Markup Language (SBML). In this study, we provide an overview of PBPK models and demonstrate how SBML, COMBINE standards, and FAIR principles can facilitate model development, coupling, and reuse. |
2023-05-28
Preprint: Eleven Strategies for Making Reproducible Research and Open Science Training the Norm at Research Institutions
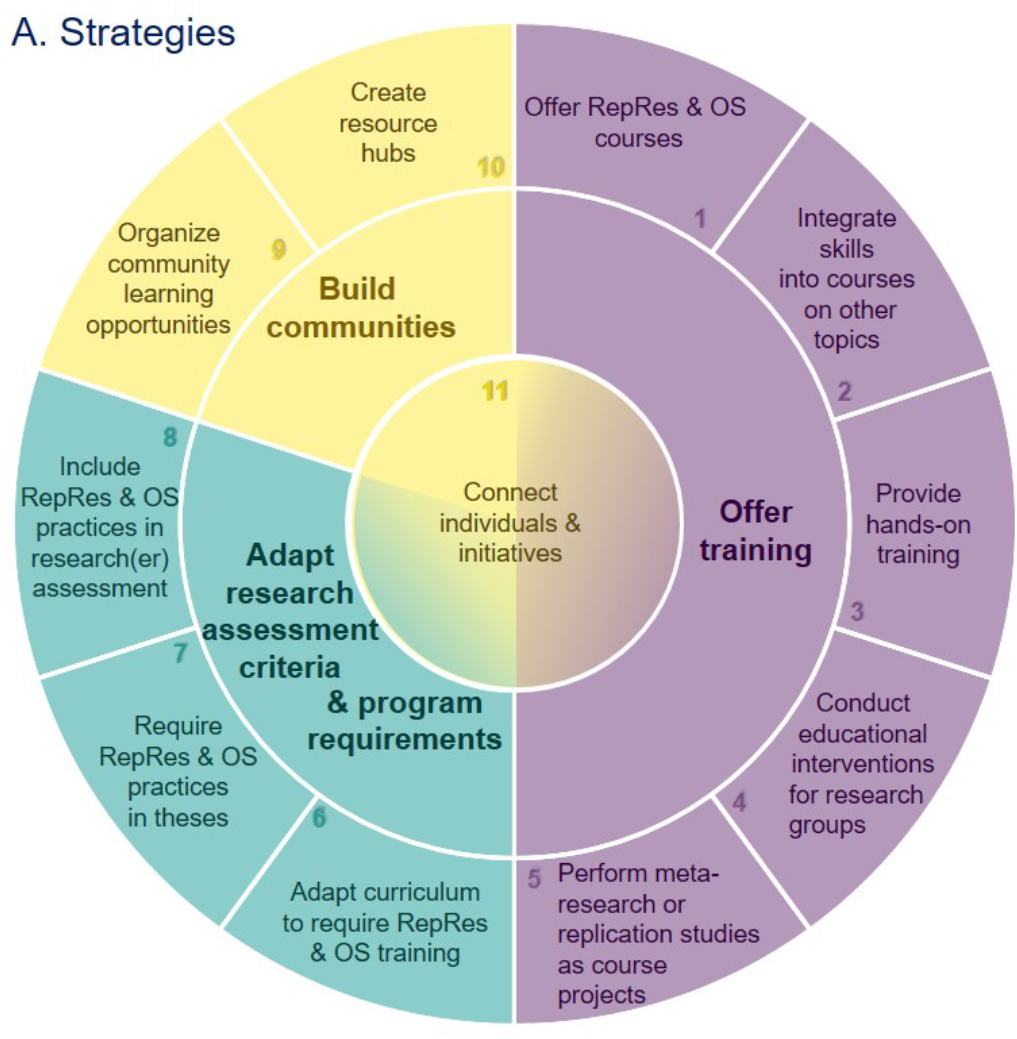
|
We present 11 strategies for making #OpenScience & #ReproducibleResearch the norm at research institutions, with tips for implementation & resources. Let's join hands to make these practices standard in the scientific community! https://www.biorxiv.org/content/10.1101/2023.04.12.536571 Across disciplines, researchers increasingly recognize that open science and reproducible research practices may accelerate scientific progress by allowing others to reuse research outputs and by promoting rigorous research that is more likely to yield trustworthy results. While initiatives, training programs, and funder policies encourage researchers to adopt reproducible research and open science practices, these practices are uncommon in many fields. Researchers need training to integrate these practices into their daily work. We organized a virtual brainstorming event, in collaboration with the German Reproducibility Network, to discuss strategies for making reproducible research and open science training the norm at research institutions. Here, we outline eleven strategies, concentrated in three areas: (1) offering training, (2) adapting research assessment criteria and program requirements, and (3) building communities. We provide a brief overview of each strategy, offer tips for implementation, and provide links to resources. Our goal is to encourage members of the research community to think creatively about the many ways they can contribute and collaborate to build communities, and make reproducible research and open science training the norm. Researchers may act in their roles as scientists, supervisors, mentors, instructors, and members of curriculum, hiring or evaluation committees. Institutional leadership and research administration and support staff can accelerate progress by implementing change across their institutions. |
2023-04-16
Preprint: A physiologically based pharmacokinetic model for CYP2E1 phenotyping via chlorzoxazone
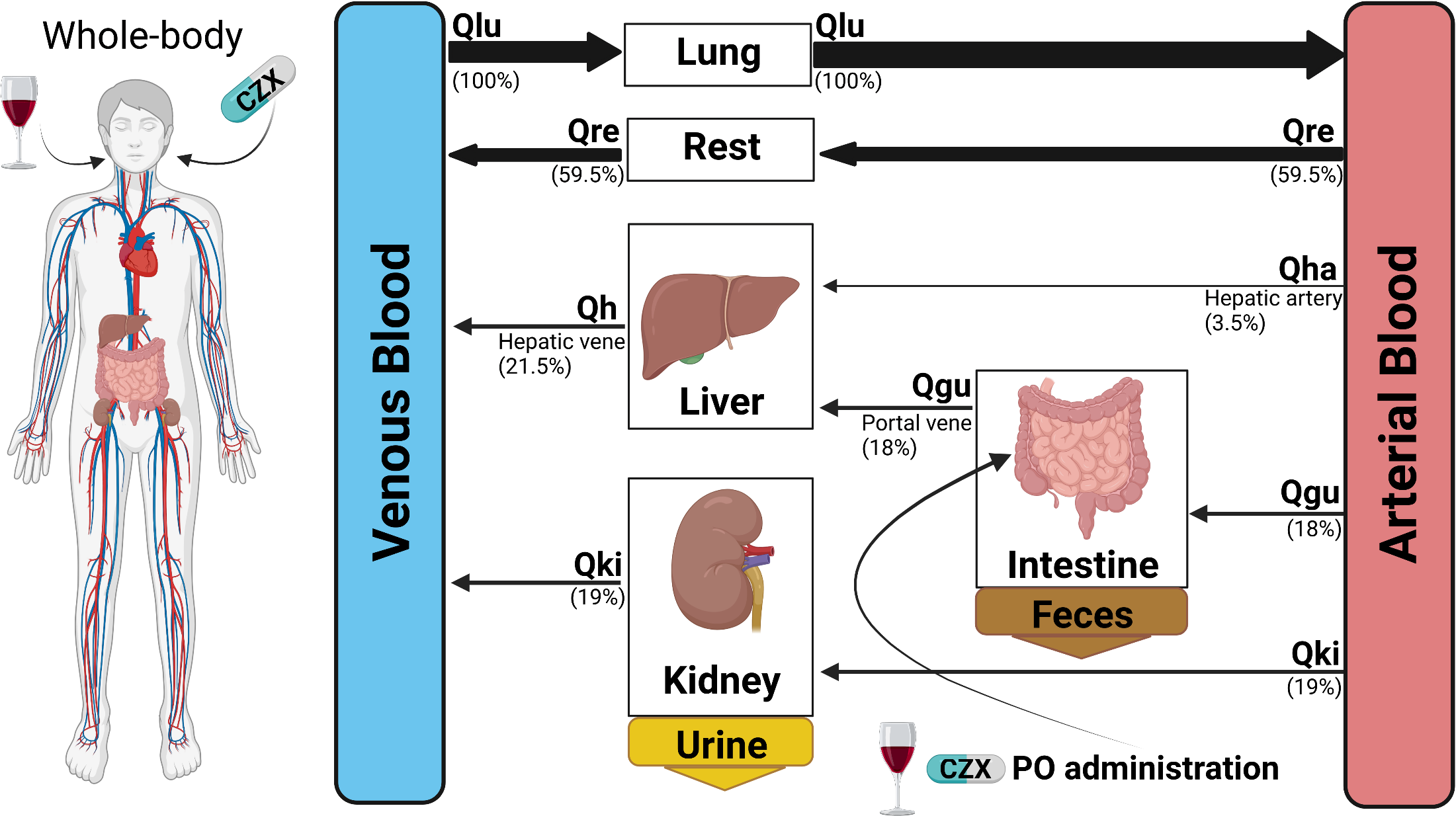
|
Our latest preprint is now on bioRxiv: A physiologically based pharmacokinetic model for CYP2E1 phenotyping via chlorzoxazone https://www.biorxiv.org/content/10.1101/2023.04.12.536571 We have developed a physiologically based pharmacokinetic (PBPK) model for CYP2E1 phenotyping via chlorzoxazone. This model takes into account various factors that can influence the activity of CYP enzymes, such as genetics, diet, age, environmental factors, and disease, making it a highly effective and reliable tool for assessing the in vivo activity of the CYP2E1 enzyme. What is truly exciting about this research is that it incorporates the effect of ethanol consumption and liver impairment on the results of metabolic phenotyping with chlorzoxazone. This means that the model is not only accurate but also highly practical, as it takes into account factors that are commonly encountered in clinical practice. The extensive pharmacokinetic dataset for chlorzoxazone and the validated PBPK model are expected to have a significant impact on the field of pharmacokinetics, as they provide a reliable and comprehensive tool for assessing the activity of CYP2E1 in a wide range of clinical contexts. This research represents a major step forward in our understanding of the factors that influence the activity of CYP enzymes and promises to have a significant impact on the development of new drugs and the optimization of existing ones. |
2023-04-03
Michael Stifel Price to promote interdisciplinary, data-driven research

|
M. Albadry, S. Höpfl, and M. König won the QuaLiPerF Michael Stifel Price for interdisciplinary, data-driven research. https://livermetabolism.com/news
M. Albadry, S. Höpfl, and M. König won the QuaLiPerF Michael Stifel Price for interdisciplinary, data-driven research for the study |
2023-03-29
Publication: Specifications of standards in systems and synthetic biology: status and developments in 2022 and the COMBINE meeting 2022
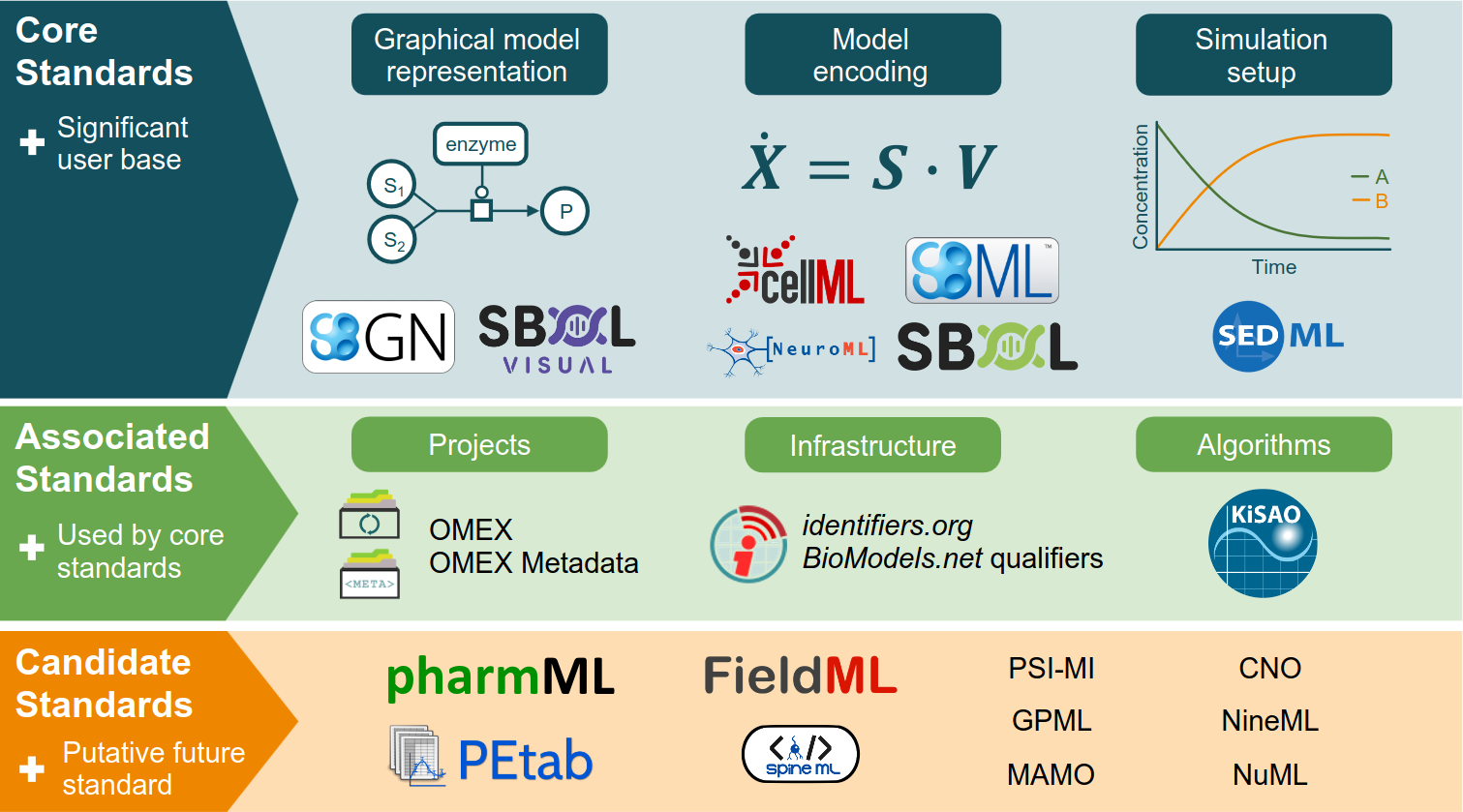
|
Our overview of specifications in systems and synthetic biology with a brief overview of the COMBINE 2022 is available. https://doi.org/10.1515/jib-2023-0004 The Journal of Integrative Bioinformatics has published our editorial for a special issue providing updated specifications of the COMBINE standards in systems and synthetic biology. The 2022 issue includes three updates to the standards, namely CellML 2.0.1, SBML Level 3 Package: Spatial Processes, Version 1, Release 1, and Synthetic Biology Open Language (SBOL) Version 3.1.0. This publication is a valuable resource for researchers looking to keep up with the latest developments in COMBINE standards. Additionally, the editorial section of the issue provides a brief overview of the COMBINE 2022 meeting held in Berlin. |
2023-03-13
Preprint: A consensus model of glucose-stimulated insulin secretion in the pancreatic beta-cell

|
Our latest preprint is now on bioRxiv: A consensus model of glucose-stimulated insulin secretion in the pancreatic beta-cell https://www.biorxiv.org/content/10.1101/2023.03.10.532028v1 The pancreas plays a critical role in maintaining glucose homeostasis through the secretion of hormones from the islets of Langerhans. Glucose-stimulated insulin secretion (GSIS) by the pancreatic beta-cell is the main mechanism for reducing elevated plasma glucose. Here we present a systematic modeling workflow for the development of kinetic pathway models using the Systems Biology Markup Language (SBML). The workflow was applied to construct the first consensus model of GSIS in the pancreatic beta-cell based on experimental and clinical data from 39 studies spanning 50 years of pancreatic, islet, and beta-cell research in humans, rats, mice, and cell lines. The model consists of detailed glycolysis and equations for insulin secretion coupled to cellular energy state (ATP/ADP ratio). Key findings of our work are that in GSIS there is a glucose-dependent increase in almost all intermediates of glycolysis. This increase in glycolytic metabolites is accompanied by an increase in energy metabolites, especially ATP and NADH. One of the few decreasing metabolites is ADP, which, in combination with the increase in ATP, results in a large increase in ATP/ADP ratios in the beta-cell with increasing glucose. Insulin secretion is dependent on ATP/ADP, resulting in glucose-stimulated insulin secretion. The observed glucose-dependent increase in glycolytic intermediates and the resulting change in ATP/ADP ratios and insulin secretion is a robust phenomenon observed across data sets, experimental systems and species. |
2023-02-12
PBPK modelling of sorafenib and Gd-EOB-DTPA

|
Frances Okibedi and Narudeem Sojimade will develop models of the anticancer drug sorafenib in HCC and the liver contrast agent Gd-EOB-DTPA.
Within this project, a high quality database of sorafenib pharmacokinetic data used in systemic therapy of hepatocellular carcinoma will be established. Based on the pharmacokinetic data set, a physiologically based pharmacokinetic (PBPK) model will be established for the evaluation of systemic sorafenib therapy in liver cancer. The model will be used to address important clinically relevant questions such as (1) sorafenib is contraindicated in advanced liver cirrhosis, (2) genetic variants of OATP uptake transporters may alter the efficacy of sorafenib, (3) CYP3A4 substrates lead to drug-drug interactions with sorafenib. Using established data curation pipelines, this project will extend our pharmacokinetics database PK-DB with literature data on Gd-EOB-DTPA. Based on the pharmacokinetic data set, a physiologically based pharmacokinetic (PBPK) model will be established for the evaluation of the pharmacokinetics of Gd-EOB-DTPA and its use as an MRI contrast agent. The model will be used to address important clinically relevant questions such as (1) the effect of renal and hepatic impairment on Gd-EOB-DTPA pharmacokinetics, (2) the effect of perfusion and OATP on Gd-EOB-DTPA pharmacokinetics, (3) the relationship between concentration and MRI attenuation. |
2023-02-12
X-Research Group - Digital twins for the treatment of hypertension
|
M. König receives funding of the Berlin University Alliance and the DFG for the X-Student Research Group: Digital twins for the treatment of hypertension
Understanding a drugs pharmacokinetics - how it is absorbed, distributed, metabolised and excreted by the body - and its pharmacodynamics - how it affects the body - is a key challenge in treating people. Angiotensin converting enzyme (ACE) inhibitors and diuretics are two classes of drugs used to treat high blood pressure. Both are among the most commonly prescribed drugs due to the high prevalence of hypertension in an ageing society. In this X-student research group we will develop physiologically based pharmacokinetic models of the diuretic hydrochlorothiazide and the ACE inhibitor lisinopril. The methodological approach is a combination of lectures, tutorials and practical work by the students. The research project is aimed at students with a STEM (science, technology, engineering, mathematics) background, including biology, computer science and medical students. Basic programming skills in Python are required. With the X-Student Research Groups, the Berlin University Alliance supports research teams consisting of young researchers and students. The goal is to involve students in current research projects of the alliance partners and to enable them to participate in (cutting-edge) research already during their studies. Funded under the Excellence Strategy of the Federal Government and the Länder by the Berlin University Alliance. |
2023-02-07
M. König has been elected as PEtab editor (2023-2026)

|
Proud to be elected editor for PEtab, a data format for specifying parameter estimation problems in systems biology. https://petab.readthedocs.io/en/latest/ PEtab is a data format for specifying parameter estimation problems in systems biology. PEtab is built around SBML and is based on tab-separated values (TSV) files. It is intended as a standardized way to provide information for parameter estimation that is beyond the current scope of SBML. This includes, for example: (1) specifying and linking measurements to models; (2) defining model outputs; (3) specifying noise models; (4) specifying parameter bounds for optimization; (5) specifying multiple simulation conditions with potentially shared parameters. Matthias König has been elected PEtab Editor from March 2023 to March 2026. |
2023-02-03
Preprint: Simvastatin therapy in different subtypes of hypercholesterolemia
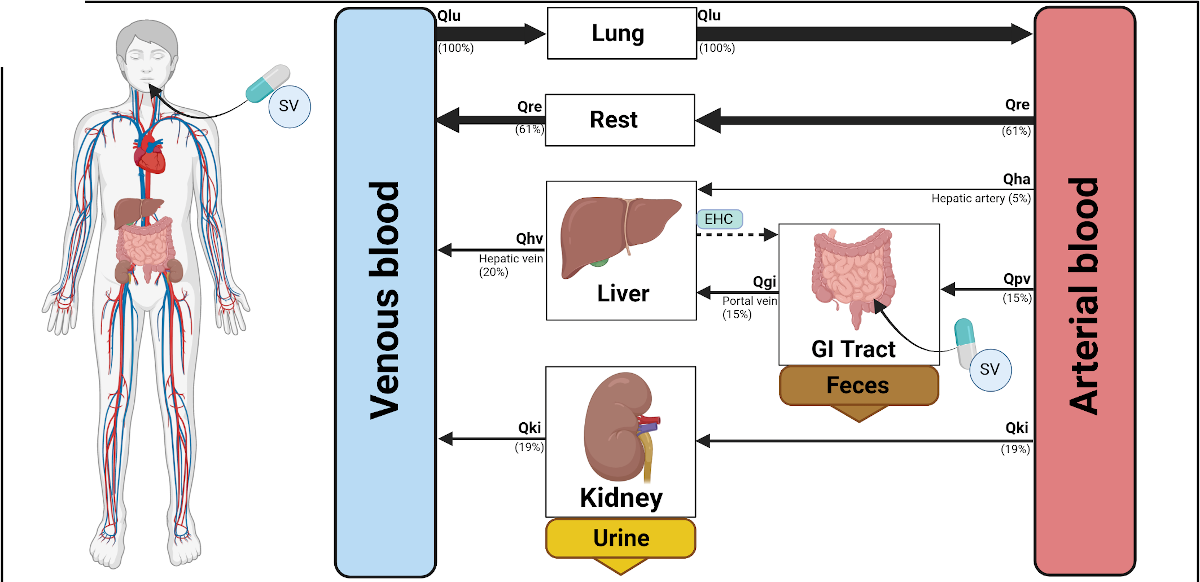
|
Our latest preprint is now on medRxiv: Simvastatin therapy in different subtypes of hypercholesterolemia - a physiologically based modelling approach. https://www.medrxiv.org/content/10.1101/2023.02.01.23285358v1 Hypercholesterolemia is a multifaceted plasma lipid disorder with heterogeneous causes including lifestyle and genetic factors. A key feature of hypercholesterolemia is elevated plasma levels of low-density lipoprotein cholesterol (LDL-C). Several genetic variants have been reported to be associated with hypercholesterolemia, known as familial hypercholesterolemia (FH). Important variants affect the LDL receptor (LDLR), which mediates the uptake of LDL-C from the plasma, apoliporotein B (APOB), which is involved in the binding of LDL-C to the LDLR, and proprotein convertase subtilisin/kexin type 9 (PCSK9), which modulates the degradation of the LDLR. A typical treatment for hypercholesterolemia is statin medication, with simvastatin being one of the most commonly prescribed statins. In this work, the LDL-C lowering therapy with simvastatin in hypercholesterolemia was investigated using a computational modeling approach. A physiologically based pharmacokinetic model of simvastatin integrated with a pharmacodynamic model of plasma LDL-C (PBPK/PD) was developed based on extensive data curation. A key component of the model is LDL-C turnover by the liver, consisting of: hepatic cholesterol synthesis with the key enzymes HMG-CoA reductase and HMG-CoA synthase; cholesterol export from the liver as VLDL-C; de novo synthesis of LDLR; transport of LDLR to the membrane; binding of LDL-C by LDLR via APOB; endocytosis of the LDLR-LDL-C complex; recycling of LDLR from the complex. The model was applied to study the effects of simvastatin therapy in hypercholesterolemia due to different causes in the LDLR pathway corresponding to different subtypes of hypercholesterolemia. Model predictions of LDL-C lowering therapy were validated with independent clinical data sets. Key findings are: (i) hepatic LDLR turnover is highly heterogeneous among FH classes; (ii) despite this heterogeneity, simvastatin therapy results in a consistent reduction in plasma LDL-C regardless of class; and (iii) simvastatin therapy shows a dose-dependent reduction in LDL-C. Our model suggests that the underlying cause of hypercholesterolemia does not influence simvastatin therapy. Furthermore, our model supports the treatment strategy of stepwise dose adjustment to achieve target LDL-C levels. Both the model and the database are freely available for reuse. |
2023-02-01
ATLAS - AI and Simulation for Tumor Liver ASsessment
|
The BMBF is funding our joint project ATLAS in the "Computational Life Sciences - AI Methods for Systems Medicine". Liver cancer is the second most common cause of cancer-related death. Diagnosis and treatment are time-critical and require highly patient-specific diagnostic and treatment pathways. Medical decision-making is based on a variety of interdependent factors related to different medical disciplines, past experience and clinical guidelines. Taking into account all decision factors in combination with the possible therapeutic approaches is a major challenge for physicians and often cannot be solved optimally even in an interdisciplinary tumor board. In this project, we are developing ATLAS, a decision support tool that will significantly assist clinicians in meeting this challenge. Based on AI methods, ATLAS processes all relevant patient data from databases, systems medicine and continuum biomechanical in silico prognosis models as well as individual patient data. The tool is being developed in a co-design approach by experts in surgical oncology, mathematical modeling and machine learning. The selected technologies will integrate automated understanding of a highly complex patient situation through simulation of liver functions with expert knowledge and ontology-driven learning with knowledge graphs from retrospective liver tumor cases. ATLAS will be based on a detailed historical data cohort of more than 6,000 patients with liver tumors and will be evaluated on case studies at the University Hospital of Jena. The integration of medical expert knowledge, mathematical modeling and artificial intelligence represents a highly original and promising approach for high-quality diagnosis and treatment of liver tumors, resulting in patient-specific improvement of prognosis. The scientific knowledge gained from these projects will provide opportunities for transfer to malignancies in other organs, such as the lung, kidney or brain. The development of tools and demonstrators will provide sustainable exploitation pathways for future commercial applications. |
2022-11-17
New Bachelor Student - Afruja Hossain

|
Afruja Hossain started working in our lab on a systematic overview of the protein variability of cytochrome P450 (CYP450) isoforms in the human liver. https://livermetabolism.com/people/
|
2022-11-15
Visiting EU parliament and attending STOA AI session

|
We were selected to participate in an European policy journey on the topic EU research funding and science communication by the Berlin University Alliance (BUA). https://www.berlin-university-alliance.de/news/items/2022/221017-bruessel-reise.html
|
2022-10-26
Humboldt Internship Program 2023 - Computational Modeling of Drug Detoxification - A Systems Medicine Approach

|
We offer Internships in 2023, application open from 01 Nov - 09 Dec 2022. https://hic.hu-berlin.de/en/internship-program/projects/201
|
2022-10-24
Dextromethorphan publication
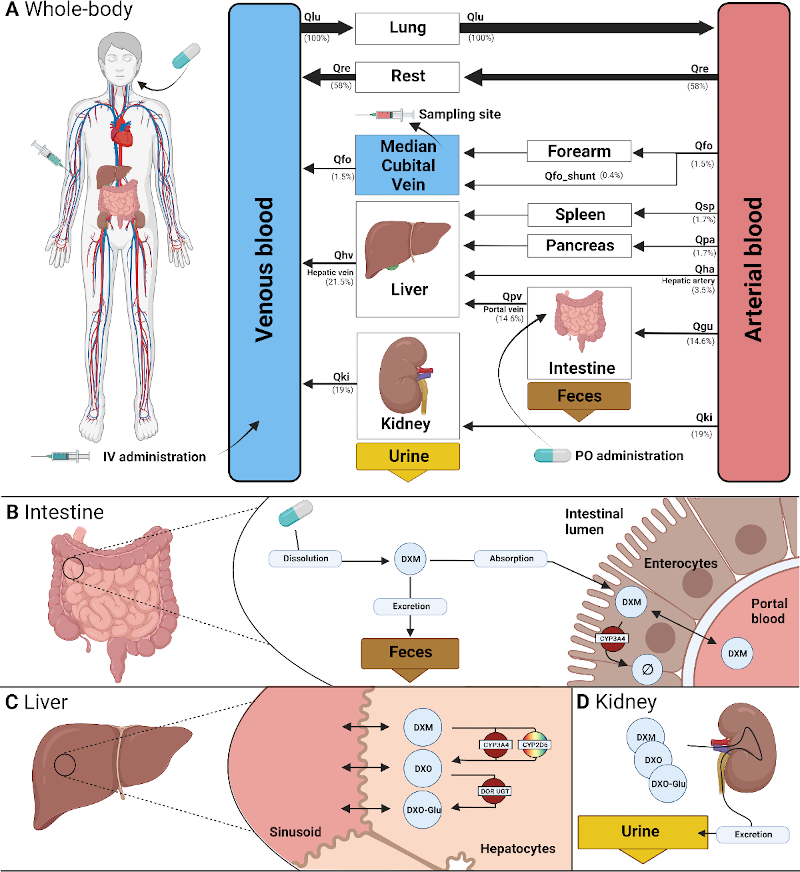
|
How does CYP2D6 genotype effect metabolic phenotyping? See our latest paper in Frontiers in Pharmacology for answers. https://www.frontiersin.org/articles/10.3389/fphar.2022.1029073/full
|
2022-10-24
X-Research Group

|
M. König receives funding of the Berlin University Alliance and the DFG for the X-Student Research Group: Physiologically based modeling of drugs: ACE inhibitors
|
2022-10-06
COMBINE 2022


|
We organized the Computational Modeling in Biology Network (COMBINE) meeting in Berlin this year with > 100 participants from 06-08 October 2022.
|
2022-10-10
ICSB 2022 - Presentation and Posters
|
We could present our work on physiologically based pharmacokinetics (PBPK) modeling at ICSB2022.
|